The interrelationship of emergency medicine and climate change and how to prioritize sustainability.
Marine Envenomations
Written by: Michael Tandlich, MD (NUEM ‘24) Edited by: Chloe Renshaw, MD (NUEM ‘22)
Expert Commentary by: Justin Seltzer, MD (NUEM ‘21)
Expert Commentary
An excellent post by Drs. Tandlich and Renshaw. Marine envenomations are common problems around the world. Like with land-based envenomations, the venomous organisms of note vary with geography; jellyfish encountered in Australia are different from those encountered in Florida, for example. As a result, we will focus on major envenomations in the United States.
The invertebrates account for a large but ultimately unknown number of envenomations. Cnidaria includes jellyfish, hydrozoa, anemones, and fire coral. A majority of stings from this group result in painful dermatitis; tentacles create a “whip-like” pattern on the skin, whereas fire coral creates localized skin wheals. The sea nettle and Portuguese man-of-war are of greatest interest, given their potential to cause severe systemic symptoms. Box jellyfish are rare in US coastal waters but produce a life-threatening toxicity.
Initial treatment is somewhat controversial. Many resources advocate for the use of seawater for the initial decontamination, given concern for vinegar triggering nematocyst release in some species common to US waters. However, further research is needed to determine which is best. At this time, seawater is recommended for empiric decontamination in the US unless a box jellyfish is strongly suspected, in which case vinegar is appropriate (a very rare circumstance). Systemically ill box jellyfish envenomations should be treated with pain and blood pressure control. The antivenom is not readily available in the US and is unlikely to be beneficial in the time course it would take to obtain it.
Echinodermata, which includes sea urchins, have mild venom on their spines that can cause local tissue irritation and pain. There are reports of severe envenomations with systemic symptoms, but this is ultimately quite rare. These injuries respond well to hot water immersion. Imaging and local wound exploration for retained spines are recommended. Soaking the wound in vinegar may help dissolve superficial spines.
Of the vertebrates, stingrays and spiny fish are of primary concern.
Stingrays stings are common and can cause serious penetrating trauma but envenomation mainly produces localized pain and swelling. The venom is heat-labile, so significant pain relief can be achieved with hot water immersion. Stingrays stings have the potential for both retained stinger and wound infections; evaluation for retained stinger with radiographs and local wound exploration is recommended along with prophylactic antibiotics.
Spinyfish, in particular stonefish, lionfish, and scorpionfish, have venom located in their spines. Stonefish have the most potent venom of any known fish. Lionfish are not native to the US but have become an invasive species. Human contact with these fish occurs both in the wild and in aquariums. These fish also have heat-labile venom susceptible to hot water immersion. However, systemically ill stonefish envenomations should receive the antivenom as this envenomation can be life-threatening; the antivenom will likely work against other spiny fish too, however, these other envenomations are usually much less severe and rarely require more than hot water immersion and supportive care.
So key learning points:
Most marine envenomations involve heat-labile venom. Hot water immersion is likely to help reduce local symptoms.
Systemic illness is rare but some marine envenomations can produce life-threatening toxicity. Be very wary of a systemically ill envenomation and try to figure out the source due to the limited availability of antivenoms.
Prophylactic antibiotics are recommended for stingray stings as they tend to get infected but otherwise are generally not necessary in most populations. Good wound care, evaluation for retained foreign bodies, and tetanus prophylaxis are the mainstays.
For further information, see this review article
Justin Seltzer, MD
UCSD Health Toxicology Fellow
Emergency Physician, UCSD Health
How To Cite This Post:
[Peer-Reviewed, Web Publication] Tandlich, M. Renshaw, C. (2022, Mar 7). Marine Envenomations. [NUEM Blog. Expert Commentary by Seltzer, J]. Retrieved from http://www.nuemblog.com/blog/marine-envenomations
Other Posts You May Enjoy
Stingray Stings
Written by: Mike Tandlich, MD (NUEM ‘24) Edited by: Peter Serina, MD, MPH (NUEM ‘22)
Expert Commentary by: Mike Macias, MD (NUEM ‘17)
Expert Commentary
Thank you Drs. Tandlich and Serina for this excellent infographic summarizing stingray envenomation! The good news is that the majority of stingray injuries are nonfatal and will heal without any complications! You hit all of the key points however I just wanted to highlight a few management tips below:
Treat as a Trauma!
While majority of the pain from stingray envenomation occurs as a result of its venom, it is important to remember that this is also a traumatic injury. Treat the injury just like you would any other penetrating trauma. Consider the location as well as surrounding structures and make sure to properly examine for tendon, nerve, and vascular injury. Injuries to the chest or abdominal regions should prompt advanced imaging and trauma consultation.
Hot Water is Key!
Stingray envenomation is noted to cause severe pain that is often out of proportion to your examination findings. While the exact mechanism is not clear, the venom can lead to not only pain but also local tissue necrosis. The good news is the venom is heat labile! The faster you can get the injured area into hot water the better. You want the water to be as hot as tolerable without causing a thermal burn. A good rule of thumb is to have the patient place their unaffected limb in the water first to see if it is tolerable. As this often occurs at a beach, lifeguards are often your best resource to get hot water fast! Oral analgesics can be administered if needed however often they are unnecessary as soon as the injured area is submerged in hot water.
Retained Barb?
While uncommon, a retained barb from the envenomation can occur so be sure to consider this and evaluate appropriately. Traditionally, x-ray imaging of the affected area is performed to evaluate for a radio-opaque barb however some evidence suggests this to be a relatively low yield practice [1]. Ultrasound can also be considered if there is suspicion for retained barb or other material. In general ultrasound has been shown to be highly sensitive for identification of foreign body [2]. Not only can it be used to identify the barb but it can be used to facilitate removal [3].
Give Prophylactic Antibiotics
Prophylactic antibiotics are recommended for stingray envenomation given that the limited data suggest a higher rate of wound infection in patients who were not initially treated with antibiotics [1]. Given these injuries often occur in the ocean make sure to cover for salt water species such as Vibro. Levofloxacin is my go to option.
Teach The Stingray Shuffle!
Keeping these key management points in mind, the good news is that the majority of stingray injuries are nonfatal and will heal without any complications! Before your patient is discharged don’t forget to remind them that the next time they are going out for a surf to do the stingray shuffle!
References
Clark RF, Girard RH, Rao D, Ly BT, Davis DP. Stingray envenomation: a retrospective review of clinical presentation and treatment in 119 cases. J Emerg Med. 2007 Jul;33(1):33-7
Aras MH, Miloglu O, Barutcugil C, Kantarci M, Ozcan E, Harorli A. Comparison of the sensitivity for detecting foreign bodies among conventional plain radiography, computed tomography and ultrasonography. Dentomaxillofac Radiol. 2010;39(2):72-78. doi:10.1259/dmfr/68589458
Nwawka OK, Kabutey NK, Locke CM, Castro-Aragon I, Kim D. Ultrasound-guided needle localization to aid foreign body removal in pediatric patients. J Foot Ankle Surg. 2014;53(1):67-70. doi:10.1053/j.jfas.2013.09.006
Michael Macias, MD
Systems Clinical Ultrasound Director,
Emergent Medical Associates
Ultrasound Director,
UHS SoCal MEC Residency Programs
How To Cite This Post:
[Peer-Reviewed, Web Publication] Tandlich, M. Serina, P. (2021, Nov 15). Stingray Stings. [NUEM Blog. Expert Commentary by Macias, M]. Retrieved from http://www.nuemblog.com/blog/stingray-stings
Other Posts You May Enjoy
TPA in Frostbite
Written by: Patrick King, MD Edited by: Nery Porras, MD (NUEM ‘21) Expert Commentary by: Anne Lambert Wagner, MD
TPA in Frostbite
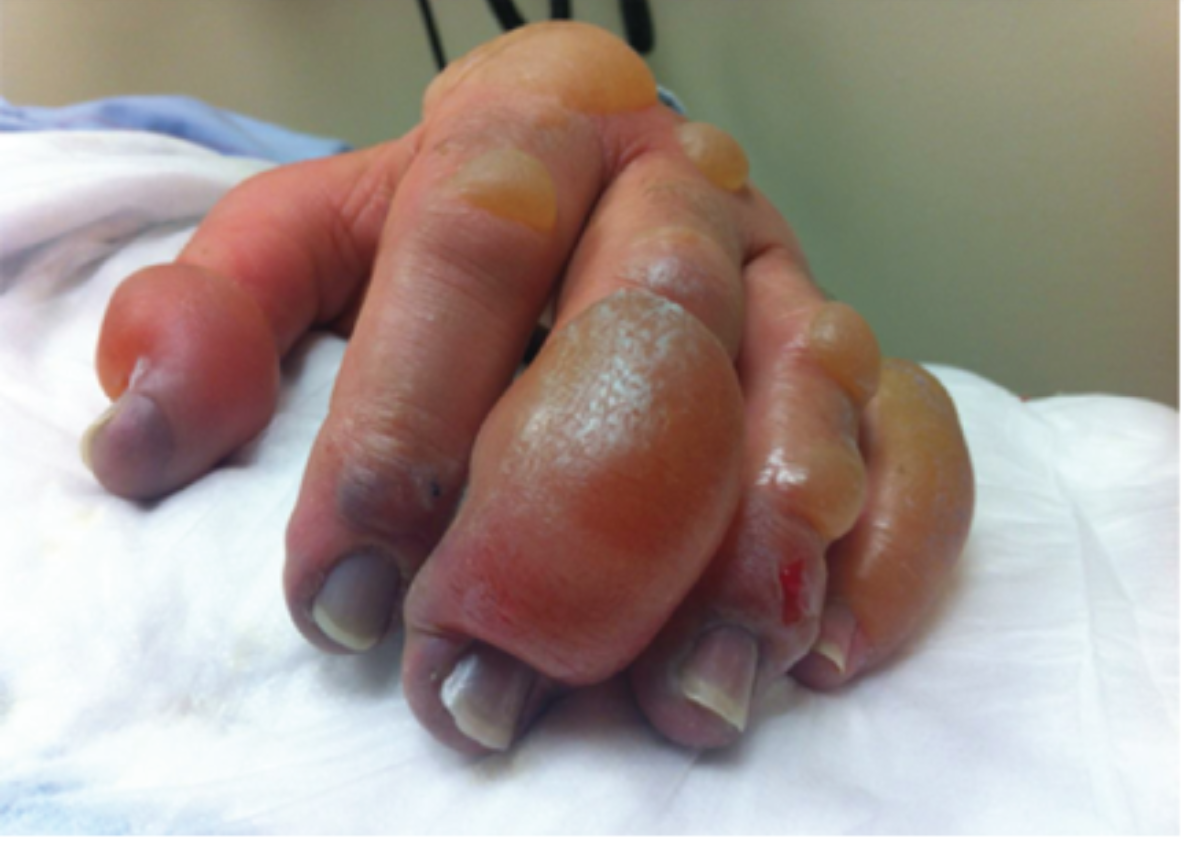
Figure 1. What we would like to avoid (Cline et al.)
It’s an early Saturday morning, and EMS brings in one of your ED’s regulars – a schizophrenic, undomiciled gentleman named Jack who finds occasional work as a day laborer. You walk to bed three to greet Jack who is uncomfortable and shivering while nursing collects vitals. His chief complaint is hand and foot pain. You listen to him speak, but you jump right into a cursory exam as he does – and your heart sinks when you see the icy hard, cyanotic, mottled digits across all four extremities. You wonder what else you might be able to offer in addition to the standard cold injury approach we are taught as emergency residents, and you recall that the What’s New in Emergency Medicine section of UpToDate just recognized growing evidence for yet another off-label use for tPA: severe frostbite.
As we head into the winter months, emergency physicians will continue to see frostbite wreck a significant level of morbidity on our most vulnerable patients – patients who are undomiciled, suffering from addictions or mental illness, and those with preexisting conditions that limit blood flow to extremities (Zafren and Crawford Mechem). This post will address the theory, evidence, and logistics behind tPA utilization in severe frostbite.
The proposed efficacy of tPA in frostbite is related to cold-induced thrombosis. Endothelial damage is sustained both as a direct result of cold-related injury and exacerbated by reperfusion injury during the period of rewarming. During rewarming, arachidonic acid cascades promote vasoconstriction, platelet aggregation, leukocyte sludging, and erythrostasis which further promote thrombosis throughout affected tissues. This process is compounded in instances of multiple freeze-thaw cycles (Cline et al).
Research on tPA in frostbite goes back years. In 2005, Twomey et al. demonstrated in an open-label study that technetium (Tc)-99m scintigraphy (i.e., nuclear bone scan) reliably predicts digits/limbs at risk for amputation. Historical control patients with no or minimal flow distal to radiographically identified “cutoff” points of ischemia on bone scans inevitably all required amputations. Untreated historical controls without flow cutoffs were more likely to retain digits. In contrast, 16 of 19 study patients with identified flow cutoffs responded to intra-arterial (IA) or intravenous (IV) tPA with an amputation rate of only 19% of at-risk digits. In 2017, Patel et al. showed a 15% amputation rate for severe frostbite in eight IA tPA patients compared to 77% in their control group.
Figure 2. Pre-tPA and Post-tPA using technetium (Tc)-99m scintigraphy bone scan (Twomey et al.)
While study results have been impressive in instances of small sample sizes such as the above, a paucity of evidence has prevented widespread utilization of tPA for frostbite use amongst emergency physicians. This year, however, What’s New In Emergency Medicine on UpToDate gave special attention to a 2020 systematic review of 16 studies by Lee and Higgins which wielded a sample size of 209 patients with 1109 digits at high amputation risk. The study, entitled “What Interventional Radiologists Need to Know About Managing Severe Frostbite”, ultimately demonstrated a 76% salvage rate amongst IA tPA (222 amputations amongst 926 digits) and 62% salvage rate in IV tPA (24 amputations amongst 63 patients). Importantly, the 16 studies are not randomized, though several such as Patel et al. and Twomey et al. utilize historical controls. There is also no direct comparison of IA vs. IV tPA, and for unclear reasons, the salvage rate for IA is in terms of digits salvaged out of those at risk while IV is expressed as a function of patients who required no amputations. Though there remains additional research to be done, UpToDate’s Frostbite authors Zafren and Crawford Mechem now give an overall grade 2C recommendation for tPA use in severe frostbite for patients otherwise at risk of life-altering amputations.
Figure 3. Grading severity of frostbite after rewarming (Cauchy et al.)
Figure 4. Grade 4 Frostbite, best seen in far right (Pandey et al.)
TPA utilization in frostbite is straightforward. UpToDate authors recommend tPA consideration for any patients with frostbite in multiple digits in a single limb, in multiple limbs, and/or in proximal limb segments who present within 24 hours of injury. The American Burn Association, which has its own guidelines (largely similar), recommends tPA for patients with cyanosis proximal to the distal phalanx after rewarming (i.e. grade 3 or 4). In more simple terms – injuries expected to be life-altering, as revealed following rapid rewarming, are likely to meet inclusion. Contraindications include general tPA contraindications as well as frostbite-specific considerations such as multiple freeze-thaw cycles which destroy tissue viability via repeated reperfusion injury as discussed previously. An additional frostbite-specific quandary with tPA use is the intoxicated frostbite patient, as substance abuse is a strong risk factor for frostbite, but intoxication can preclude tPA consent.
So you suspect you have a candidate – how do you proceed? Advice from UpToDate’s Zafren and Mechem is representative of many experts’ approaches. Early consultation with centers experienced in advanced frostbite therapeutics is recommended. General immediate frostbite care is undertaken on ED arrival, including 15-30 minutes rapid water bath rewarming at 37 to 39 degrees Celsius, at which point the tissue should change from hard and cold to more soft and pliable. Ensure adequate analgesia, as this rewarming process can be painful. Following rapid rewarming, the grade of frostbite can be assessed (fig. 2,3). Clinical suspicion is then confirmed via technetium (Tc)-99m scintigraphy (bone scan) or by angiography at centers with expertise in intra-arterial tPA use. Angiography is utilized only if IA administration is planned. UpToDate recommends IV tPA for most candidates given the ease of administration unless specific institutional protocol differs.
Specific UpToDate dosing regimen is as follows: “Give a bolus dose of 0.15 mg/kg over 15 minutes, followed by a continuous IV infusion of 0.15 mg/kg per hour for six hours. The maximum total dose is 100 mg. After tPA has been given, adjunct treatment can be started with IV heparin or subcutaneous (SC) enoxaparin. The dose of IV heparin is 500 to 1000 units/hour for six hours or targeted to maintain the partial thromboplastin time (PTT) at twice the control value. Enoxaparin can be given at the therapeutic dose (1 mg/kg SC).”
Additional research remains to be done on this topic. At this time, however, it is reasonable to give your patients – a hand – when it comes to severe frostbite. Consider tPA.
Expert Commentary
Background
Skin and soft tissue are readily susceptible to injury at either end of the temperature spectrum. With exposure to cold, unprotected tissues can readily become frostbitten and/or hypothermic (aka Frostnip); two distinct but often linked injuries. In the past, skin, limbs, and digits sustaining severe frostbite injury had predictable outcomes: sloughing or amputation. The only question was how long to wait to amputate. Essentially no progress was made in the treatment of frostbite until the early 1990’s when the development of a treatment protocol for frostbite patients was developed using thrombolytics to restore blood flow to damaged tissue.
Frostbite has two separate mechanisms to the injury itself. The initial insult is the cold injury that leads to direct cellular damage from the actual freezing of the tissues. Rewarming of the affected tissues leads to the second, a reperfusion injury resulting in patchy microvascular thrombosis and tissue death.
Figure 1. Frostbite
Frostbite Classification
First-degree frostbite: Superficial damage to the skin from tissue freezing with redness (erythema), some edema, hypersensitivity, and stinging pain.
Second-degree frostbite: Deeper damage to the skin with a hyperemic or pale appearance, significant edema with clear or serosanguinous fluid-filled blisters, and severe pain. Frostnip, first and second-degree frostbite will generally heal without significant tissue loss.
Third-degree frostbite: Deep damage to the skin and subcutaneous tissue. Cold, pale, and insensate without a lot of tissue edema. Shortly after rewarming, edema rapidly forms along with the presentation of hemorrhagic blisters. Significant pain often occurs after rewarming.
Fourth-degree frostbite: All the elements of a third-degree injury with evidence of damage extending to the muscle, tendon, and bone of the affected area.
Figure 2. 1st and 2nd degree frostbite (left), 3rd and 4th degree frostbite (right)
Pre-hospital or Emergency Department Management
Determining the extent of frostbite injury starts with a detailed history regarding how the affected area appeared on presentation.
The history of a cold, white, and insensate extremity on presentation is consistent with severe frostbite injury (3rd and/or 4th-degree frostbite).
A severe frostbite injury requires emergent therapy with thrombolytics unless the patient meets one of the exclusion criteria.
If in question regarding the depth of the injury, a clinical exam can be supported by a vascular study as indicated. A digital Doppler exam is a simple and quick modality to further Clarify the diagnosis of severe frostbite.
Complete a primary survey to rule out any traumatic injuries.
Correct hypothermia (warm room, remove wet clothing & jewelry, warmed fluids, etc.)
If there are areas of frozen tissue rapid rewarming is preferred (see next section, rapid rewarming is associated with the best outcomes and salvage rates. However, never thaw until the risk of re-freezing has been eliminated. Patients undergoing freeze-thaw cycles do not respond to thrombolytics and are treated with standard supportive frostbite therapy.
Protect affected areas from further trauma with padding, splinting, and immobilization while transporting.
Keep the patient non-weight bearing to avoid incurring additional injury to frozen tissue (ice crystals) and/or disrupting blisters.
Elevate the affected extremities when able to decrease tissue edema.
Obtain a large-bore peripheral IV & start warmed fluids. Most patients will present with dehydration secondary to hypothermia and/or intoxication.
Avoid direct radiant heat to prevent iatrogenic burns to the cold tissue.
Update the patient’s tetanus status
Expect the patient to have increasing pain as the involved tissue is rapidly rewarmed. Pain management should include scheduled Ibuprofen (800 mg if no contraindication) to block the arachidonic cascade, gabapentin (nerve pain), and narcotics as needed.
Figure 3. Rewarming
Rapid rewarming
Circulating water bath when able. Put each affected area in its own water bath to avoid the tissue “knocking” against each other.
Document start & completion time
Try to keep the water temp at 104 ºF (40º C)
It will take 30-45 min for a hand or foot
If the patient has boots, socks, gloves, etc frozen to the skin do not force off. Submerge the entire area as part of the rapid rewarming process
Continue until frostbitten limb becomes flushed red or purple, and tissue soft and pliable to gentle touch
Air Dry
Avoid any aggressive manipulation to decrease tissue loss and injury
Elevate the affected areas to decrease swelling
Dress the affected areas with bulky padded dressings for transfer to avoid trauma to the areas
Avoid rewarming with a direct heat source (heat lamp, warm IV bag, etc.). This will lead to a thermal injury secondary to the lack of blood flow.
Rewarming will be associated with:
A return of sensation, movement, and possible initial flushing of the skin. The vessels in the case of severe frostbite (3rd or 4th degree) quickly become thrombotic (<20 minutes) with mottling or demarcation, however, the demarcation may be subtle at first and requires careful observation.
In the case that the tissues return fully to a normal color and palpable pulses or Doppler digital signals are present, the patient may not need any further intervention other than close observation (inpatient or daily visits in the clinic) and pain management.
If any question exists, an urgent triple-phase bone scan can support perfusion to the affected area.
Figure 4. Early evidence of demarcation and patchy thrombosis
Indications for Thrombolytics
Patient presenting with frozen tissue (severe frostbite, 3rd and/or 4th degree)
Absent or weak Doppler pulses following rewarming
Clinical exam consistent with severe frostbite
< 24 hours of warm ischemia time (time from rewarming)
Time matters significantly. For each hour after rewarming delaying the start of thrombolytics decreased salvage rates even by 28.1%.
With correct training after discussion with a burn center that does a lot of frostbite care, thrombolytics can be safely started at the outside hospital prior to transfer to the center.
Frostbite Thrombolytic Protocol
Examine for any associated injuries or illnesses. If any question of injury the patient will require a head, chest, and abdominal CT to rule out any sources of bleeding.
The dosing of the thrombolytic requires an actual weight and while infusing the thrombolytic requires ICU status and monitoring for 24 hours.
Following completion of the therapy, the patient will immediately be started on treatment dose Enoxaparin for 1-2 weeks.
Figure 5. Patient before and after receiving thrombolytics
Contraindications to the Thrombolytic Protocol
Absolute contraindications:
> 24 hours of warm ischemia time
Repeated freeze/thaw cycles
Concurrent or recent (within 1 month) intracranial hemorrhage, subarachnoid hemorrhage or trauma with active bleeding
Inability to consistently follow a neurologic exam (eg. intubated and sedated, significant dementia)
Severe uncontrollable hypertension
Relative contraindications:
History of GI bleed or stroke within 6 mo.
Recent intracranial or intraspinal surgery or serious head trauma within 3 months
Pregnancy
Figure 6. Clinical guide for the management of frostbite
Frostbite Take-Home Points
Rapid rewarming of frozen tissue in a circulating water bath is the preferred method of rewarming.
Patients that have undergone trauma in conjunction with the frostbite injury are not an absolute contraindication to receiving tPA.
Starting tPA at the outside hospital, prior to transport, results in significantly improved outcomes even compared to those that receive it at UCH.
Frostbite patients, regardless of whether or not they get thrombolytics, do better at a center that has experience and protocols to take care of frostbite.
Anne Lambert Wagner, MD, FACS
Associate Professor
University of Colorado
Medical Director
Burn & Frostbite Center at UC Health
How To Cite This Post…
[Peer-Reviewed, Web Publication] King, P. Porras, N. (2021, Aug 16). TPA in Frostbite. [NUEM Blog. Expert Commentary by Lamber Wagner, A]. Retrieved from http://www.nuemblog.com/blog/TPA-in-frostbite.
Other Posts You Might Enjoy
References
Cauchy E, Davis CB, Pasquier M, Meyer EF, Hackett PH. A New Proposal for Management of Severe Frostbite in the Austere Environment. Wilderness & Environmental Medicine. 2016;27(1):92-99. doi:10.1016/j.wem.2015.11.014.
Cline D, Ma OJ, Meckler GD, et al. Cold Injuries. In: Tintinalli's Emergency Medicine: a Comprehensive Study Guide. New York: McGraw-Hill Education; 2020:1333-1337.
Grayzel J, Wiley J. What’s New in Emergency Medicine. In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. (Accessed on November 3, 2020.)
Lee J, Higgins MCSS. What Interventional Radiologists Need to Know About Managing Severe Frostbite: A Meta-Analysis of Thrombolytic Therapy. American Journal of Roentgenology. 2020;214(4):930-937. doi:10.2214/ajr.19.21592.
Pandey P, Vadlamudi R, Pradhan R, Pandey KR, Kumar A, Hackett P. Case Report: Severe Frostbite in Extreme Altitude Climbers—The Kathmandu Iloprost Experience. Wilderness & Environmental Medicine. 2018;29(3):366-374. doi:10.1016/j.wem.2018.03.003.
Patel N, Srinivasa DR, Srinivasa RN, et al. Intra-arterial Thrombolysis for Extremity Frostbite Decreases Digital Amputation Rates and Hospital Length of Stay. Cardiovascular and Interventional Radiology. 2017;40(12):1824-1831. doi:10.1007/s00270-017-1729-7.
Twomey JA, Peltier GL, Zera RT. An Open-Label Study to Evaluate the Safety and Efficacy of Tissue Plasminogen Activator in Treatment of Severe Frostbite. The Journal of Trauma: Injury, Infection, and Critical Care. 2005;59(6):1350-1355. doi:10.1097/01.ta.0000195517.50778.2e.
Wagner A, Orman R. Frostbite, Asystole, Perfectionism, EQ, Middle Way, Flu. January 2019 - Frostbite - Frostbite, Asystole, Perfectionism, EQ, Middle Way, Flu | ERcast. https://www.hippoed.com/em/ercast/episode/frostbite/frostbite. Published 2019. Accessed November 3, 2020.
Zafren K, Crawford Mechem C. Frostbite: Emergency Care and Prevention. In: UpToDate, Post TW (Ed), UpToDate, Waltham, MA. (Accessed on November 3, 2020.)
Health Risks Imposed by the Beach
Written by: Alex Herndon, MD (NUEM ‘21) Edited by: Ashley Amick, MD, MS (NUEM ‘18) Expert Commentary by: Patrick Lank, MD
With warm weather fast approaching, it’s time to break out the sunscreen and beach gear. Besides protecting oneself from UV rays and heat exhaustion, there are other dangerous pathogens lurking in the sandy shores that are worth being aware of as patients begin to flood the Emergency Department during summer vacation. Here are a few dangerous diseases to consider that masquerade as common chief complaints.
1. More than just swimmer’s itch
A 17 year old female presents to the Emergency Department complaining of a patchy skin rash that developed only a couple days after her first swim of the summer. Freshwater lakes house trematode parasites that upon contact leads to cercarial dermatitis, otherwise known as “swimmer’s itch”. Symptoms typically develop 2 days after exposure and last a week. Relief can be easily obtained with antihistamines and corticosteroid cream. [1]
Now consider that same patient is returning from a trip from Key West, Florida for a Bachelorette Party. You notice that her legs are shaved and there are several small nicks around her ankles. She is presenting with a worsening red rash on her lower leg that is red, warm, blistering, and in some locations has formed superficial ulcers. While it can be easy to chalk this us to severe sunburn and possible superimposed cellulitis, it is important not to miss this deadly necrotizing skin infection caused by Vibrio vulnificus, commonly known as one of many “flesh-eating bacteria.” Unlike the more benign trematode, V vulnificus can be found in brackish or saltwater, and in North America most commonly in the Gulf of Mexico. [2] V vulnificus infects wounds and leads to skin breakdown and ulceration and if not treated immediately infection has a mortality rate of anywhere from 25-50%. [3] Given the virulence of the disease, it is important to treat early and aggressively. The mainstay treatment for V vulnificus includes intravenous 3rd generation cephalosporins along with a tetracycline such as doxycycline. Source control becomes prudent and may require surgical debridement. [4]
2. “It’s just a cough”
With the warm weather finally here, a 60 year-old retiree began breaking in his paddle board along the shores of Lake Michigan. To cool off afterwards, he would hit the public beach showers. One week later he shows up at the Emergency Department complaining of body aches, low grade fevers, and a cough that won’t quit. While the bacteria Legionella pneumophila is typically associated with hot tubs, don’t forget other warm freshwater places this microbe loves to grow, including beach showers, air-conditioning units, and outdoor misters like those seen at amusement parks and sporting events. [4] People fall ill after inhaling aerosolized droplets from the contaminated sources.
Pontiac fever is a mild form of Legionella infection, presenting as vague flu-like symptoms that typically resolve in 2 to 4 days without treatment. However, the more severe form of infection, commonly known as Legionnaires disease, presents as pneumonia with cough, fever and myalgias. Unlike other bacterial pneumonias, Legionnaires is also more commonly associated with gastrointestinal symptoms like nausea, vomiting, and diarrhea, and can also cause hyponatremia. On average 15% of cases per year have been fatal, thus never forget to start atypical coverage for pneumonia, such as azithromycin, which provides adequate coverage for Legionella infection. [5] And if Legionella is diagnosed or highly suspected, alerting local health authorities is important because early containment of possible sources, such as public showers, is imperative to preventing a deadly outbreak.
3. Beyond febrile seizures
A 10 year-old boy is sent to sailing camp in Wisconsin. While he was well upon arrival, after only 3 days his parents get a call that their son has been hospitalized. His camp counselors brought him to the ED after he became febrile and had a seizure a day after capsizing in the lake. They reported throughout the day the boy had been complaining of a headache and was increasingly lethargic. Typically the constellation fever, headache, altered mental status, and seizure heralds bacterial meningitis. However given this child’s unique summer camp experience, one must consider other environmental exposures that pose a risk.
While rare, warm freshwater lakes can house the deadly Naegleria fowleri, more commonly known as “the brain-eating amoeba.” [6] This amoeba enters via the swimmer’s olfactory nerve, reaching the brain where it causes primary amebic meningoencephalitis (PAM). Patients present within 1 day to 2 weeks after exposure, first with flu-like symptoms including fever, headache and vomiting, that eventually progress to involve hallucinations and seizures. Similar to any patient presenting with symptoms concerning for meningitis, performing a lumbar puncture is key in making the diagnosis. N fowleri can be identified within cerebral spinal fluid either via direct visualization, antigen detection or PCR. While the majority of cases have been fatal, with a fatality rate of nearly 98%, survival is possible if identified and treated early with miltefosine, an anti-leishmania drug. [7]
During these warm summer months it is vital to understand where your patients have been and what they have been doing because knowing those details can end up saving their lives.
Expert Commentary
Thank you Drs. Herndon and Amick for these wonderful reminders that there are more things to be afraid of at the beach than sharks (and/or Sharknados). While this blog post contains great tidbits on three diagnoses, I think these cases also highlight times when a careful focused clinical history changes the emergent work-up and treatment. These patients could have easily been diagnosed with another condition and had their definitive care delayed, so thank you for these reminders.
As a native Floridian who grew up within walking distance of the Atlantic Ocean, I think there are a few additional entities for the emergency physician to consider when treating beachgoers. My medical toxicology training is begging me to direct this commentary towards my wheelhouse, but I will resist and will be sure to mention some other diagnoses.
But to start, I have to bring up intoxication. For those readers who do not live in the Midwest of the United States, I want to make you aware that Chicago has a wonderful series of beaches. Having been working in an emergency department in Chicago for 15 years now, I also have to point out that the number one reason patients are brought to the ED from a beach is for alcohol intoxication. Higher temperatures, increased thirst, increased physical activity, prolonged drinking, and possible co-ingestion of other mind-altering substances all increase the chances that a day at the beach will end in the ED. So be careful, warn your teenage/twenty-something family members, and consider checking an ethanol concentration in altered beachgoers.
The geographic proximity our ED has to the beach and Lake Michigan also means we see a lot of drownings. Some are intentional, others accidental; some are associated with traumatic injuries, others with intoxication; some patients are pediatric, some are geriatric. Despite their variations, all drowning should be taken seriously and involve aspects of resuscitation that are worth reviewing when you get a chance. Although it is now a few years old, I recommend reading the review article “Drowning” by D Szpilman, et al. from NEJM in 2012 (DOI: 10.1056/NEJMra1013317). It’s a great review with some helpful references for people interested in reading more.
Finally I would recommend anyone working in a clinical environment where the weather is about to turn warmer should review the clinical features and resuscitation of patients with heat-related injuries and superficial burns. When I was a PGY-1 in Chicago and had my first patient check in with a sunburn, I was in complete shock. Why did this person not know homecare for a sunburn? Easy, I thought: lots of aloe, move like a mummy for a day, and bathe in self-loathing and regret. But years of experience in a northern clime have taught me that changes in seasons are particularly dangerous for these injuries – people are out of practice, they forget, or they simply don’t care. No matter the reason, these early parts of the season are when we see big upticks in significant presentations.
In summary, thank you again for bringing up these infectious complications of having fun at the beach. But if you want to scare some sense into your 15-year-old nephew, don’t only tell him about Naegleria fowleri – please also terrify him with stories of overdoses, drowning, and severe hyperthermia.
Patrick Lank, MD, MS
Assistant Professor of Emergency Medicine
Medical Toxicologist
Department of Emergency Medicine
How To Cite This Post:
[Peer-Reviewed, Web Publication] Herndon, A. Amick, A. (2021, Mar 15). Health Risks Imposed by the Beach. [NUEM Blog. Expert Commentary by Lank, P]. Retrieved from http://www.nuemblog.com/blog/health-risks-imposed-by-the-beach.
Other Posts You May Enjoy
References
Parasites: Cercarial Dermatitis. Centers for Disease Control and Prevention. 2012 January. <https://www.cdc.gov/parasites/swimmersitch/faqs.html>
Thompson, H. Eight diseases to watch out for at the beach: “Flesh-Eating” bacteria. The Smithsonian. 2014 August. <https://www.smithsonianmag.com/science-nature/diseases-watch-out-beach-18095234 6/>
Horseman, M. Surani, S. A comprehensive review of Vibrio vulnificus: an important cause of severe sepsis and skin and soft-tissue infection. Int J Infectious Diseases. 2011 March: 15(3): 157-166.
Thompson, H. Eight diseases to watch out for at the beach: Pontiac Fever and Legionnaires Disease. 2014 August. <https://www.smithsonianmag.com/science-nature/diseases-watch-out-beach-180952346 /#mftUupdDj 5cwE00L.99>
Healthy Swimming: Respiratory Infections. Centers for Disease Control and Prevention. 2016 May. <https://www.cdc.gov/healthywater/swimming/swimmers/rwi/respiratory-infections.html>
Thompson, H. Eight diseases to watch out for at the beach: “Brain-Eating” Amoeba. The Smithsonian. 2014 August. <https://www.smithsonianmag.com/science-nature/diseases-watch-out-beach-180952346 /#mftUupdDj5cwE00L.99>
Parasites: Naegleria fowleri - Primary Amebic Meningoencephalitis - Amebic Encephalitis. Centers for Disease Control and Prevention. 2017 February. <https://www.cdc.gov/parasites/naegleria/pathogen.ht ml>
Altitude Illness
Written by: Alex Herndon, MD (NUEM ‘21) Edited by: Danielle Miller, MD (NUEM ‘19) Expert Commentary by: Gabrielle Ahlzadeh, MD
Expert Commentary
During my four years of residency at sea level, I never treated a patient with altitude sickness. Now, living in Utah and working at a ski clinic where the peak is just over 11,000 feet, I see it almost weekly. Patients tend to be surprised when we diagnose them with acute mountain sickness, either because they are physically fit, otherwise healthy or have been to altitude before and never had symptoms. Educating patients that altitude sickness can affect anyone, regardless of how many marathons they’ve run, is important in ensuring that they follow directions to manage their symptoms. A lot of patients also don’t realize that it takes a few days to develop altitude sickness, and that days 2-3 are usually when symptoms develop. Oftentimes, not sleeping well may be the first symptom. If patients present with symptoms of poor sleep and headaches, it’s important to instruct patients to take it easy and take time to adjust, as well as the importance of staying hydrated and doing their best to get enough sleep. It’s helpful to frame this as days lost on the mountain so patients take their mild symptoms seriously.
Anecdotally, most patients improve pretty rapidly with oxygen administration so when any patient from out of town presents with vague symptoms, our first step in ski clinic is to put them on oxygen . Some patients look pale and ill while others don’t even look sick, and you’re often shocked by their low oxygen saturation. We’ve had fit young patients with oxygen saturations in the 70s who look completely fine, which again, just stresses the importance of obtaining vitals and not being fooled by healthy and fit patients. I’ve seen kids who present with fatigue, vomiting and headache who look sick and then after an hour of oxygen and some fluids, bounce right back to their normal selves.
Obviously it’s important to maintain a broad differential for patients who present with symptoms of altitude sickness, while recognizing that it is a diagnosis that can tie together multiple symptoms. This is especially true in pediatric patients who cannot articulate their symptoms clearly. Checking an initial blood sugar is part of our initial workup, especially in kids. But, if you don’t consider acute mountain sickness, then you won’t be able to make your patient feel better with oxygen, descent or other medications.
From the ski clinic, we often send patients home with portable oxygen tanks mainly to use while they are sleeping, since poor sleep often makes symptoms worse. We treat most patients with both acetazolamide and dexamethasone and frequently recommend they come back to clinic the next day for reassessment. We often recommend that patients sleep at lower altitude and just come up for skiing if possible. For patients with evidence of pulmonary edema, they must descend and are sent to the ER for closer monitoring and treatment. The same would be true with any patient with evidence of altered mental status.
Gabrielle Ahlzadeh, MD
Clinical Assistant Professor of Emergency Medicine
University of Southern California
How To Cite This Post:
[Peer-Reviewed, Web Publication] Herndon, A. Miller, D. (2020, Aug 31). Altitude Illness. [NUEM Blog. Expert Commentary by Ahlzadeh, A]. Retrieved from http://www.nuemblog.com/blog/altitude-illness
Other Posts You May Enjoy
Management of Environmental Heat Injury in the ED
Written by: Sean Watts, MD (NUEM PGY-3) Edited by: Phil Jackson, MD (NUEM ‘20) Expert Commentary by: George Chiampas, DO, CAQSM, FACEP
Heat related illness has become an increasing source of morbidity and mortality due to environmental injuries from rising global temperatures and increased interest in outdoor activities. The National Oceanic and Atmospheric Administration reported that 2016 was the hottest year on record, and that temperatures were on average 3.2 F° higher than the 20th century averages.[1] Increasing temperatures have manifested in fatal heat waves such as one claiming the lives of 70,000 individuals living in Europe during 2003.[1] The population most subject to these heat waves include the extremes of age and athletes.
Human body temperature is normally set at 37 ° C, and is maintained via the preoptic nucleus of the anterior hypothalamus.[1,2] Hyperthermia results from exposure to an exogenous heat source without altering the hypothalamic set point. As core temperatures elevate during exertion and with exposure to heat, the posterior hypothalamic nucleus signals sympathetic pathways that result in vasodilation of peripheral vascular beds and shunting blood away from gastrointestinal vasculature in order to maximize heat dissipation. Additionally, eccrine sweat glands are cholinergically activated resulting in an evaporative cooling effect. When the duration and magnitude of heat exposure outpace these physiologic mechanisms, the symptoms of heat-related illness become evident and vary from mild heat cramps to severe heat stroke and death.[2]
Heat cramps result from both potassium wasting from persistent utilization of aldosterone in order to maintain a euvolemic state and sodium loss through sweat. Edema can result from increased hydrostatic pressure of the peripheral vasculature. Additionally, syncope and hypotension can manifest due to dehydration, orthostatic pooling of blood, peripheral vasodilation, and a subsequent decrease in cardiac output. Without appropriate treatment, heat exhaustion and the more extreme heat stroke can present.
Heat exhaustion is defined as a core temperature between 37 ° C and 40 ° C with signs and symptoms including intense thirst, weakness, discomfort, anxiety and dizziness.[1,2,6,8] Heat stroke, on the other hand, is defined as a core temperature greater than 40 C° with signs of central nervous system dysfunction. Heat stroke can be further categorized into exertional and non-exertional.[4] The demographic of exertional heat stroke includes athletes, military personal, or young individuals participating in prolonged exercise.[4,8] Non-exertional heat stroke includes the elderly, young children, or individuals with metabolic or cardiac comorbidities that engage in brisk to minor activity at elevated temperatures.[1,4] When the body reaches 40 C° denaturation of proteins, release of pro-inflammatory mediators, and direct activation of the coagulation cascade occurs.[1,2] This can ultimately result in disseminated intravascular coagulation, which is a common complication of heat stroke.[1,4,5] Disruption of the liver and the cerebellum from tissue ischemia, hypoxia, vascular dysfunction, secondary cascade inflammation manifest with elevated liver function tests and ataxia dysmetria, and coma.[1,6]
Summary of the Pathophysiology of Heat Stroke [1]
Treatment of heat related illness in the emergency department rests on appropriate recognition of the severity of disease. For heat syncope and heat cramps, isotonic or hypotonic electrolyte solutions may be administered in addition to actively flexing leg muscles to prevent peripheral pooling of blood.[7,8] Ice packs or cold towels around the neck, axillae and groin can also be used for comfort measures 6. In general, these heat illnesses are self-limiting.
For heat exhaustion and heat stroke, treatments become more aggressive and should be initiated within 30 minutes of recognition of the signs/symptoms.[1,4] These patients often present critically ill and rapid assessment of the patient’s airway, breathing, and circulation is paramount. Caregivers should obtain good IV access, as well as intubate the patient if they are obtunded or in danger of loss of airway protection.[1,6] Broad spectrum critical care labs should be obtained, as well as a CK to assess for evidence of rhabdomyolysis.[5] Additionally, obtaining an accurate core body temperature is a crucial first step to determine the severity of illness.[1,2,4,5,6] This is best performed through continuous rectal probe monitoring. Rehydration should then be performed, preferably with 1 to 2 L of isotonic fluids.[1,4] Care should be taken to not over-correct hypovolemia as the aforementioned pathophysiology makes this population vulnerable to pulmonary edema.[1] Additionally, care should be taken not to over bolus hypotonic or isotonic solutions as this population, especially those involved in long distance endurance sports like triathlons or marathons, are particularly prone to hyponatremia.[9] If these patients are given too much of these solutions, this can actually exacerbate the hyponatremia. Patients with profound hyponatremia will actually require IV hypertonic solutions or salt tabs.[9]
Clinicians should next focus on cooling core body temperature. The best treatment for exertional heat stroke is cold-water immersion therapy—where the patient gets placed in a cold body of water.[5,7] This method takes advantage of the high thermal conductivity of water and is most effective when the patient’s clothing is removed. Studies have demonstrated that immersion in an ice-water slurry at 2°C generated cooling rates of 0.35°C/min.[4] Comparatively, allowing hyperthermic subjects to rest in air-conditioned or temperature-controlled rooms only resulted in cooling rates of only 0.03°–0.06°C/min.[4] Evidence regarding an optimal temperature to halt cooling is still under debate, but is thought to be somewhere between 38°C to 39°C, with the fear that overcooling may result in cardiac arrhythmias, especially in the elderly suffering from non-exertional heat stroke.[1,4]
Subject in a cold water-immersion bath after heat- stroke [4]
The use of cold-water immersion therapy in non-exertional heat stroke is still under debate, but the limited evidence shows that evaporative and convective cooling by a combination of cool water spray with continual airflow over the body may be superior, especially in the elderly suffering from non-exertional heat stroke.[4] In many emergency departments, complete cold water immersion therapy may not be readily available and limited by the placement of cardiac leads, intubation, and IV access, so evaporative and convective cooling methods become first-line for both exertional and non-exertional heat stroke in the emergency department setting should cold water immersion be unavailable.[1,4,5] Should shivering become problematic, benzodiazepines are considered first line therapy.[1,6] In severe or refractory cases the patient may benefit from ECMO.[6]
Evaporative and conductive cooling methods--note the placement of ice packs in axilla, groin as well as the cooling fan overhead [4]
With the rapid increase in heat-related injuries, and projected increase in global warming, researchers are continually seeking new and efficacious treatments. For example, recombinant activated protein C is currently being explored to manage the disseminated intravascular coagulation that may result from heat stroke.[2] Additionally, application of cold packs versus other methods of rapid cooling has been explored. An experimental study published in the journal of Wilderness and Environmental Medicine found that the use of ice packs provided a significantly higher enthalpy change over cold packs—suggesting that ice packs are more efficacious than cold packs when managing heat-injury.[3] Additionally, the study found that application of cold packs or ice packs to locations high in AV anastomoses provided superior cooling rates.[3] Evaporative plus convective cooling units are also under study as an alternative means to cold water immersion for the treatment of non-exertional heat stroke.[5]
Key Points and Summary
Heat Injury continues to be a major cause of environmental morbidity and mortality, and will likely increase due to rising global temperatures
Heat Injury exists on a continuum, with heat cramps/syncope on one end and heat exhaustion/stroke on the other end
Obtain a rectal temperature if you suspect heat exhaustion/stroke, assess ABC’s, get good IV access, and be careful not to over bolus isotonic/hypotonic solutions due to the risk of worsening hyponatremia in athletes
If feasible, cold water immersion is superior for exertional heat stroke, in the ED setting evaporative and conductive cooling with ice packs can be used
In severe or resistant cases cardiopulmonary bypass can be effective
table of cooling methods [6]
Expert Commentary
A great review and reminders of what is a preventable death especially in exertional heatstroke. Unfortunately, still in the United States there are still approximately fifteen to twenty heat-related deaths in athletes annually, mostly seen in august. While there is a spectrum of illness, preventative measures, a high index of concern and management can all mitigate negative outcomes.
In non-exertional heat illness, removal from the environment, addressing the medical condition and or removing any contributing factors is key. Cooling methods and the aggressiveness of cooling are determined by the patient’s mental status and stability. As highlighted, Heat exhaustion presents with headaches, nausea, dizziness, and weakness. Using cooling blankets and cold packs to the groin axilla and circulating fans all are measures in passive cooling. One key element to address as typically a patient presents undifferentiated is to obtain a rectal temperature in a timely fashion as highlighted. Temperatures and glucose in altered mental status patients are critical for efficient management and positive outcomes. There are key studies that highlight that time and duration above 42C lead to higher morbidity including death.
In athletics, the death of Minnesota football player Korey Stringer in August of 2001 shed greater light on the risks of exertional heatstroke. Since his death, more work and research has been done including best practices in sport to mitigate these outcomes. Across many sports, including Marathons, best practices as outlined in the blog are being implemented pre-hospital. These measures are comparable to the recent out of hospital cardiac arrest best practices of on-sight CPR and utilization of an AED and transport second mantra. In heatstroke “cooling” on sight with ice tub submersion is the current thread being communicated. This messaging is evidenced by a recent EMS consensus paper that highlights to first-responders the importance of recognizing but also cooling on-sight prior to transport. The delay of cooling and transport times to delay of recognition and cooling in emergency departments may lead to not initiating life-saving rapid cooling beyond the thirty minutes highlighted in the blog.
As you accurately highlighted patients can present differently, however, the key is altered mental status (AMS). Based on experience this can have the forms of patients collapse and obtunded, seizing, irritable and combative to just being confused. Rapid assessments in the right environment with excluding other AMS possibilities will allow the practitioner to respond and manage in a timely fashion. At Northwestern, both Dr. Malik and Dr. Chiampas have published the attached “collapse algorithm” (below) which allows for a quick assessment and possible differential diagnoses. Lastly obtaining a rectal temperature, which at times may be challenging with the combative patient, allows the staff in the Emergency room to objectively determine when to cease cooling. I will share that some of these patients based on their presentation would traditionally be intubated upon arrival. I would caution and remind the practitioner that if you have prepared in advance and can rapidly cool the symptoms are reversible within 10-15 minutes of ice submersion.
Lastly for emergency departments, where out-door events (sporting, festivals or concerts) with the possibility of stimulant use, preparedness is key. At Northwestern, we have secured 100-gallon ice tubs, implemented the collapse algorithm in our trauma bay and on when high-risk events take place to trigger necessary resources. For the Chicago Marathon, Triathlon or major concerts such as Lolla Palooza we order ice to the ER, towels, and prep the tub while educating our staff of the likelihood of these conditions. As we head towards the summer ahead with all of the environmental concerns of climate change and increased temperatures, this blog provides key reminders of the emergency department’s role.
George Chiampas DO CAQSM
Assistant Professor Northwestern University, Feinberg School of Medicine
Departments of Emergency and Orthopedic Surgery
Chief Medical Officer U.S. Soccer
Chief Medical and Safety Officer Bank of America Chicago Marathon
Team Physician Chicago Blackhawks
How To Cite This Post
[Peer-Reviewed, Web Publication] Watts S, Jackson P. (2020, July 6). Management of Environmental Heat Injury in the ED [NUEM Blog. Expert Commentary by Chiampas G. Retrieved from http://www.nuemblog.com/blog/environmental-heat-injury
Other Posts You May Enjoy
References
Heat-Related Illness. Walter F. Atha, MD. Emerg Med Clin N Am 31 (2013) 1097–1108. http://dx.doi.org/10.1016/j.emc.2013.07.012
Wilderness Medical Society Practice Guidelines for the Prevention and Treatment of Heat-Related Illness: 2014 Update. Grant S. Lipman, MD; Kurt P. Eifling, MD; Mark A. Ellis, MD; Flavio G. Gaudio, MD; Edward M. Otten, MD; Colin K. Grissom, MD. WILDERNESS & ENVIRONMENTAL MEDICINE, 25, S55–S65 (2014)
Chemical Cold Packs May Provide Insufficient Enthalpy Change for Treatment of Hyperthermia. Samson Phan, MS; John Lissoway, MD; Grant S. Lipman, MD. WILDERNESS & ENVIRONMENTAL MEDICINE, 24, 37–41 (2013)
Cooling Methods in Heat Stroke Flavio G.Gaudio MD∗Colin K.Grissom MD†The Journal of Emergency Medicine, Volume 50, Issue 4, April 2016, Pages 607-616
Heat Stroke. Alan N. Peiris, MD, PhD, FRCP(London); Sarah Jaroudi, BS; Rabiya Noor, BS. JAMA. 2017;318(24):2503. doi:10.1001/jama.2017.18780
Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 8e. Judith E. Tintinalli, J. Stephan Stapczynski, O. John Ma, Donald M. Yealy, Garth D. Meckler, David M. Cline. Section 16, Chapter 210: Heat Emergencies. http://accessmedicine.mhmedical.com.ezproxy.galter.northwestern.edu/content.aspx?bookid=1658§ionid=109384117. Accessed June 10, 2019.
Heat-Related Illness in Athletes Allyson S. Howe MD, Barry P. Boden, MD First Published August 1, 2007 https://doi-org.ezproxy.galter.northwestern.edu/10.1177/0363546507305013
Heat-Related Illnesses. ROBERT GAUER, MD, Womack Army Medical Center, Fort Bragg, North Carolina. BRYCE K. MEYERS, DO, MPH, 82nd Airborne Division, Fort Bragg, North Carolina. Am Fam Physician. 2019 Apr 15;99(8):482-489.
Hyponatremia among runners in the Boston Marathon. Almond CS, Shin AY, Fortescue EB, Mannix RC, Wypij D, Binstadt BA, Duncan CN, Olson DP, Salerno AE, Newburger JW, Greenes DS. N Engl J Med. 2005 Apr 14;352(15):1550-6.
Marathon: The Collapsed Athlete
Written by: Zach Schmitz, MD (PGY-3) Edited by: Andrew Berg, MD (NUEM ‘19) Expert Commentary by: Jake Stelter, MD
Marathon: The Collapsed Athlete
You’ve been enjoying a beautiful, 71 degree day wrapping ankles and rehydrating runners at the last marathon medical tent, just one mile from the finish. Suddenly, you get a different, more concerning type of call - there’s a runner down about a block south.
You fight against the flow of runners and finally see your patient on the left side of the course. He’s laying on his back and a bystander has placed an ice bag on his head. He tells you his name is Tony, but can’t tell you where he is or what he was doing. A friend says he was slowing down and looking unsteady before sitting on the curb. He’s sweating, working a little hard to breathe, and he has a 2+ radial pulse. What is your approach?
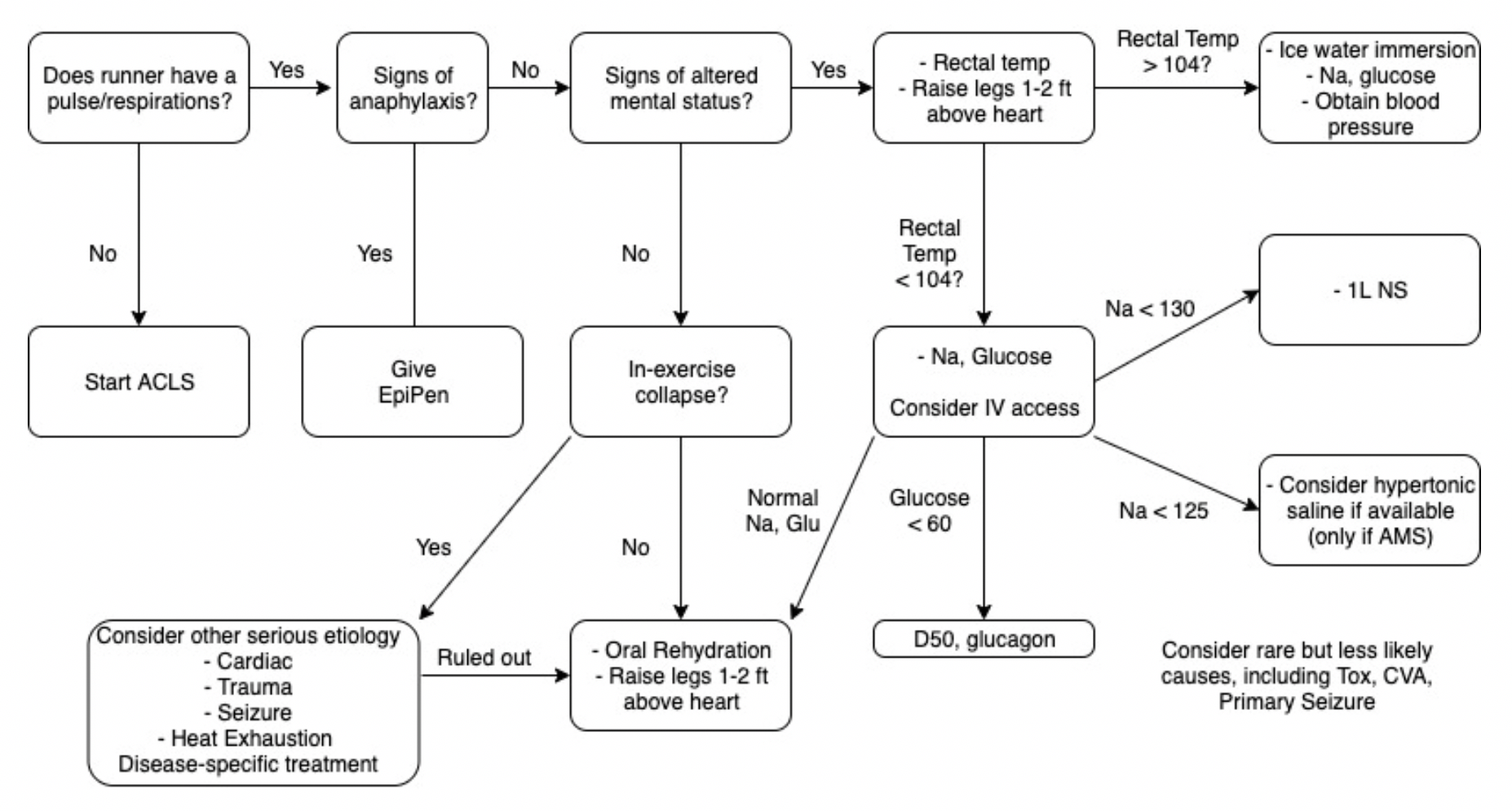
What to rule out first:
Just as with other ED patients, the first thing to do is rule out or intervene on life-threatening causes of runner’s collapse. Collapse during exercise is particularly concerning. There are five main causes of downed runners in that category: Sudden Cardiac Arrest, Exertional Heat Stroke, Anaphylaxis, Hypoglycemia, and Hyponatremia [1]. Below is an approach aimed toward addressing these concerns.
1. Sudden Cardiac Arrest
Suggested by an absent pulse and/or abnormal respirations.
Do not delay treatment. Start ACLS/BLS as your training and equipment allows and transport to a nearby ED.
Extremely rare[2]
2. Anaphylaxis
Suggested by any combination wheezes/stridor, shortness of breath, swelling, skin changes, nausea/vomiting, and altered mental status.
Treatment will likely be limited to IM epinephrine, as antihistamines, H2 blockers, and steroids are not routinely stocked in medical tents.
3. Exertional Heat Stroke
Suggested by altered mental status and a rectal temperature of > 104 degrees F.
These patients should be placed in an ice bucket immediately – any delay will risk permanent neurologic dysfunction.
Ice bucket immersion has been shown to reduce core body temperature 3x faster than ice towels and 15x faster than ice packs over major arteries[3].
Rapid on-site cooling is associated with better outcomes than immediate transfer to an emergency department for cooling. Those on site cooling end-points are controversial, but getting below 102 degrees F consistently leads to a safe transfer.[4]
You have to use rectal temperatures, as other temperature measurements have proven unreliable in a marathon setting.[4}
4. Hypoglycemia
Suggested by a spectrum from tremor, anxiety, diaphoresis, and altered mental status, up to seizure and coma.
Patients should be treated with glucose and transferred to a nearby medical facility.
5. Hyponatremia
Suggested by paresthesias, nausea/vomiting, and altered mental status, up to seizure and coma.
In one study of the Boston Marathon, 13% of runners had sodium values < 130, and 0.6% had critical values < 120. Those with longer race times, weight gain during the race, and those at the extreme ends of the BMI scale were more likely to have problems.[5]
Normal saline should be started for patients with initial Na of 130 or below, and 3% NS may be considered if Na < 125.
Thankfully, the above conditions comprise the minority of visits to medical tents at the marathon (including for downed runners). So what do you do with someone who is down and lightheaded but with a temperature of 99.3, sodium of 138, glucose of 98, and no signs of anaphylaxis? Collapse during exercise is still concerning, even after ruling out the causes above. You’ll want to confirm the patient can tolerate oral rehydration, place in a Trendelenberg position, and likely refer for further testing.
Collapse after exercise is more common, and, fortunately, often benign. Exercise associated collapse is likely to be the most frequent condition you encounter if you are in the final medical tent.
Exercise Associated Collapse (EAC)
Although considered to be more a chief complaint than diagnosis, EAC is defined as “a collapse in conscious athletes who are unable to stand or walk unaided as a result of light headedness, faintness and dizziness or syncope causing a collapse that occurs after completion of an exertional event.”[4] In one study, it accounted for 59% of patient presentations at the final medical tent.[1]
While running, increased oxygen demand by muscle leads to increased cardiac output and decreased peripheral vascular resistance. Skeletal muscle works as “second heart” for the race, overcoming this decrease in PVR to increase venous return. This mechanism is lost when running stops, and blood pools in the lower extremities. Cardiac output cannot be maintained, and perfusion is decreased. Further, the baroreceptor reflex controlling this mechanism is often compromised during long exercise. [6]
It is a fairly simple mechanism to reverse, and therefore a simple condition to treat. Placing the patient in a Trendelenberg position with the legs above the heart will usually achieve a fluid equilibrium in 10-30. Holtzhausen showed that these patients have no different electrolyte concentrations and are no more volume depleted than runners who finished the race without complication, so IV fluids are unnecessary[7]. However, keep in mind these people just ran a marathon, so they could probably use a little oral rehydration.
These patients should prove capable of sitting, then standing, then walking and eating before being discharged from the tent. If they show signs of altered mental status, vital sign abnormalities, or electrolyte imbalances, they should be treated appropriate and then transferred to an emergency department.
The vast majority of runners visiting a marathon medical tent are fully capable of finishing the race and just need help working out a cramp, covering up a blister, or grabbing some gel to cool a sore muscle. However, serious conditions do happen, and it is important to keep them in mind the next time you volunteer at your local marathon.
Take away points:
Life-threatening pathology is certainly possible in this relatively healthy cohort
Sudden cardiac arrest, exertional heat stroke, anaphylaxis, hypoglycemia, and hyponatremia should be considered for every down runner with altered mental status
Approach to the down runner: make sure you don’t need ACLS on scene > transfer to tent for rectal temp > Na, Glucose
If rectal temp is > 104, go directly to ice bath. Fully cool before transferring patient from tent
Although syncope post race can be scary, EAC is likely to resolve with 10-30 minutes of raised legs and oral rehydration
Expert Commentary
This is a great review of managing marathon runners who are acutely ill. It is important to keep in mind the diagnoses pointed out when dealing with a collapsed athlete.
1. Sudden Cardiac Arrest: This should be treated promptly following BLS/ACLS protocols. In this situation, the goal is to get to early defibrillation if possible as the most common cause is going to be a shockable arrhythmia, either ventricular fibrillation or ventricular tachycardia. The resources immediately available to you will vary depending on where on the course the patient goes down. Early activation of EMS is critical as they will bring with them both the means of transportation as well as ACLS supplies to aid in resuscitation.
2. Altered Mental Status: In a marathon athlete, the most important and life-threatening cause of altered mental status that needs to be ruled out is exertional heat stroke. As correctly pointed out, a core rectal temperature should be obtained on any athlete that is altered. Once identified as having a core temp over 103F, the athlete should be immediately cooled in an ice water tub until their temperature is 102F. At this point, the athlete should be removed from the water. Cooling below 102F can cause rebound hypothermia as cool peripheral blood shunts to the core. Avoid starting IV’s in runners prior to cooling, as getting blood into the tubs will contaminate them. If the athlete is normothermic and altered, check for hypoglycemia and treat accordingly.
3. Hyponatremia: Exercise-Induced Hyponatremia (EIN) is a relatively rare but very serious complication of endurance events. It is generally caused by excess sodium loss (sweating) that is often accompanied by excess free water intake. In a patient that is having signs and symptoms of neurologic dysfunction that is normothermic and not hypoglycemic, consider EIN. Common signs and symptoms include paresthesias, confusion, muscle weakness, cramping and seizures. If you have the ability to check a rapid sodium, then you can treat accordingly. If the patient’s sodium level is below 130 WITHOUT neurological symptoms, restrict free water intake and consider oral rehydration with electrolyte solutions. Sparingly administer isotonic IV fluids, no more than 250-500mL at a time and recheck the sodium level after each small bolus. If a patient falls into the category of hypervolemic hyponatremia, they may actually have an excess of ADH hormone and giving fluid may precipitate an even further drop in sodium. If a hyponatremic patient is having any neurological manifestation, especially seizures, the treatment is administration of 3% sodium chloride solution in 50-100mL boluses.
Also, as pointed out, be sure to consider other potential causes of your patient’s symptoms, including but not limited to cardiac pathology, trauma, stroke, and exercise associated collapse. Patients that are undifferentiated will often need to be transported to the nearest Emergency Department, as you are unlikely to have the resources to complete a diagnostic work-up in your course medical tent.
Jacob Stelter, MD
Instructor of Clinical Emergency Medicine
Primary Care Sports Medicine Fellow
University of Cincinnati
Medical Committee - Lead ICU Tent Coordinator
Bank of America Chicago Marathon
How to Cite This Post
[Peer-Reviewed, Web Publication] Schmitz Z, Berg, A. (2020, April 20). Marathon: The Collapsed Athlete. [NUEM Blog. Expert Commentary by Stelter, J]. Retrieved from http://www.nuemblog.com/blog/marathon
Other Posts You Might Enjoy:
References
[1] Roberts W, O’connor F, Grayzel J. Preparation and management of mass participation endurance sporting events. UpToDate. May 23 2017. https://www.uptodate.com/contents/preparation-and-management-of-mass-participation-endurance-sporting-events
[2] Roberts W.O., and Maron B.J.: Evidence for decreasing occurrence of sudden cardiac death associated with the marathon. J Am Coll Cardiol 2005; 46: pp. 1373-1374
[3] Casa D et al. Exertional Heat Stroke: New Concepts Regarding Cause and Care. Curr Sports Med Rep. 2012 May-Jun;11(3):115-23.
[4] Childress MA, O'Connor FG, Levine BD. Exertional collapse in the runner: evaluation and management in fieldside and office-based settings. Clin Sports Med. 2010 Jul;29(3):459-76. doi: 10.1016/j.csm.2010.03.007.
[5] Almond et al. Hyponatremia among runners in the Boston Marathon. N Engl J Med. 2005 Apr 14;352(15):1550-6.
[6] Asplund CA, O'Connor FG, Noakes TD Exercise-associated collapse: an evidence-based review and primer for clinicians Br J Sports Med 2011;45:1157-1162.
[7] Holtzhausen LM1, Noakes TD, Kroning B, de Klerk M, Roberts M, Emsley R. Clinical and biochemical characteristics of collapsed ultra-marathon runners. Med Sci Sports Exerc. 1994 Sep;26(9):1095-101.
[8] Madan S., Chung E., The Syncopal Athlete. American College of Cardiology. http://www.acc.org/latest-in-cardiology/articles/2016/04/29/19/06/the-syncopal-athlete. Apr 29 2016. Accessed 10 1 2017.
Cold Injury Management in the ED
Written by: Sean Watts, MD (PGY-2) Edited by: David Kaltman, MD (PGY-4) Expert commentary by: Pinaki Mukherji, MD
Learning Objectives:
• Recognize the signs and symptoms of freezing injury
• Understand the pathophysiology of freezing cold injury and how it manifests in the delineated signs and symptoms
• Discuss the treatment practices for managing freezing injury in the emergency department
• Discuss barriers to effective treatments for these conditions, and current research to improve outcomes
Introduction
As interests in outdoor activities and the rates of homelessness have increased over the past twenty years, so has the level of patients presenting with cold injuries. Due to this increasing prevalence, it is important for emergency physicians to understand the signs, symptoms, and management of these injuries. This is especially true for emergency physicians working in areas where mountaineering, skiing, ice-climbing and other outdoor activities are popular, or in areas with significant cold exposure and large populations of undomiciled patients.
Cold injuries get divided into two categories: non-freezing injuries and freezing injuries. Non-freezing injuries include trench foot, pernio, panniculitis, and cold urticaria and are generally due to prolonged exposure to damp, non- freezing conditions. Freezing injuries include frostbite and its associated severity classifications. Of these categories frostbite remains the most severe and, and can present in a range of symptoms from clear blisters and cyanosis, to hard non-deforming necrotic skin.
Treatment of both non-freezing cold injuries and frostbite are similar, and co-presentation often occurs. However, if a patient presents with concomitant hypothermia, this should be treated first. The course of treatment for hypothermia should be guided by a patient's level of consciousness, shivering intensity, and cardiovascular stability in the field since accurate temperature readings cannot always be obtained. A rectal temperature should be obtained in the emergency department and active rewarming measures such as heat pads, or heated humidified oxygen should be utilized, and application of these rewarming devices should be applied to the areas of the body with the potential for greatest convective heat loss—back, axilla. While rewarming takes place, the practitioner should monitor vitals and provide CPR, AED, and intubation, as necessary.
Freezing Injury
The mechanism that accounts for frostbite is due to direct cell death due cold exposure and further cell death due to ischemia. As water freezes in tissues it expands—poking holes in the cellular membrane—leading to hyperosmotic cell death. This cell death is further exacerbated by what is known as the hunting reaction—an alternating freeze/ thaw cycle due to local alternating vasoconstriction and vasodilation. Emboli form in the vasculature due to endothelial damage, resulting in ischemia, leading to destruction of the microvasculature and localized cell death.
Frostbite can occur anywhere on the body but generally occurs on the distal extremities, face, nose, and ears. The injured area often appears pale and feels stiff and cold, and patients endorse stinging and numbness.
Now, frostbite gets divided into three zones: the zone of coagulation, the zone of hyperemia, and the zone of stasis. The zone of coagulation is distal and where the cellular damage is most severe. The zone of hyperemia is superficial, proximal, and has the least cellular damage. The zone of stasis is between the two and has the most potential for intervention to salvage tissue.
Frostbite also gets characterized into four different classifications schemes based on severity of injury and prognosis of recovery. First-degree frostbite, also referred to as frostnip, is characterized by partial skin freezing with erythema, edema, and has excellent outcomes. Second-degree injury is defined by full-thickness skin freezing, formation of substantial edema, erythema, and the formation of clear blisters. Second degree injury has the most potential for intervention with modest outcomes. Third-degree injury is defined by damage that extends into the subdermis with associated hemorrhagic blisters and necrosis of skin and necrosis of skin, appearing as a blue-gray discoloration. Fourth-degree injury is characterized by further extension into adipose tissue, muscle, and bone, with little edema that forms a dry black eschar. Fourth degree frostbite tends to have the “mummified” appearance of dry gangrene.
First Degree Frostbite
Second degree frostbite--note the clear filled blisters characteristic of this degree of frostbite
Third degree frostbite--note the areas of hemorrhagic blisters, characteristic of third degree frostbite
Fourth Degree Frostbite
In the field and the emergency department, treatment should be focused on preventing refreezing injury. It is imperative that active thawing measures not be initiated unless the thawed state can be maintained (remember the hunting reaction!). Preliminary measures to help thawing include hydration, administration of low molecular weight dextran (it has been shown to reduce blood viscosity and decrease thrombi), and NSAIDS that will reduce prostaglandin and thromboxane release. If it is possible to maintain a thawed state, active thawing can take place by submersion in a water bath maintained between 37 C and 39 C. The emergency physician will know the rewarming process is complete when the affected area becomes red or purple and is soft and pliable. If the affected area is an extremity, it should be elevated in order to prevent dependent edema from forming. When rewarming occurs patients will often note severe pain, and patients should be treated with parenteral opioid therapy. Other post-thaw therapy includes antithrombotic drugs—tPa has been used widely in addition to heparin, as well as vasodilating agents. When patients present within 24 hours, with multiple digits affected, or evidence of multiple limbs affected, intra- arterial tPA can be utilized along with intra arterial heparin. Iloprost has also been suggested for grade 2-4 frostbite when patients present <48 hours after injury. If blisters form, they should be treated with topical aloe vera cream every 6 hours, and tetanus immunization status should be assessed and given if needed.
One question that commonly arises in the emergency management of frostbite is does this injury need surgery? In general, early surgical intervention is not indicated for the management of frostbite. Studies have demonstrated that early surgery contributes to unnecessary tissue loss and poor cosmetic results. This stems from the inability to assess the depth of frostbite at its early stages and that tissue below blackened necrotic tissue is regenerating. Technetium (Tc)-99m scintigraphy often gets used after maximum rewarming therapy to predict long-term viability of affected tissue. Escharotomy is the only early surgical intervention indicated if the patient has range of motion or circulation abnormalities. Most patients with frostbite can be discharged from the emergency department with good follow up--barring a situation where the individual will simply be re exposed to cold temperatures or they require admission for pain management.
Future Directions
While non-freezing and freezing injuries continue to become common occurrences in austere environments and amongst undomiciled patient populations, developmental therapies to improve outcomes continue to be researched. For example, iloprost, as been gaining popularity, as it was recently used in Sweden with success. It is a prostacyclin analogue that mimic the effects of a sympathectomy and helps to prevent emboli from forming. In several studies it has proven more effective than tPA administration; however, it is not a currently approved FDA drug. More large scale clinical trials and cohort studies are needed, as many of these trials have low sample sizes.
Summary and Pearls
Suspect non-freezing or freezing injury in undomiciled patients or in patients with prolonged exposure in cold environments
Perform a thorough neurovascular exam of the afflicted digit/extremity, and attempt to grade if consistent with frostbite
Only begin rewarming if the warm state can be maintained
Frostbite, even when severe is not a surgical emergency
Consider iloprost or tPA for appropriate candidates
Expert Commentary
This is a nice overview of the spectrum of presentation of freezing injury. I would reinforce a few key points to give practical context in the treatment of these patients.
Systemic signs always take priority when resuscitating a cold injured patient. As such, the rewarming measures described will utilize dry heat, and target mental status and cardiovascular status. In contrast, in the rewarming of freezing injury, moist heat is always preferred.
As mentioned here, non-freezing and freezing injury coexist, and are not always easy to tell apart. While non-freezing injury may be outside of the scope of this blog post, severe cases of trench foot can appear similar to 2nd or 3rd degree frostbite injuries. Blister formation in non-freezing injury is rare, but can occur, and the sloughing of skin that occurs can be mistaken for ruptured blisters.
While the tissue damage of freezing injury can be severe, the deep necrosis that results is typically dry gangrene, whose natural course is auto-amputation. While a non-freezing injury like trench foot typically has better outcomes, severe cases can leave patients with circulatory compromise and non-intact skin, leading to wet gangrene and potentially sepsis.
Rapid rewarming of freezing injury (15 to 60 min.) is supported by animal models, with immersion being the preferred modality. The author above writes that rewarming should commence “if it is possible to maintain a thawed state.” For patients who have treatment initiated in the field, or en route to comprehensive care, this is a judgment not to be made lightly, as refreezing leads to much worse tissue destruction.
NSAIDs are indicated in the treatment of freezing injury, with an excellent safety to benefit ratio. The use of other agents aimed at improving consequences of thrombosis are less certain and should be reserved for more severe cases. Both tpa and iloprost have been associated with lower amputation rates in small studies and case series, along with PGE1 and isosorbide dinitrate as potential agents showing promise.
Low molecular weight dextran is recommended by Wilderness Medicine Society guidelines, with a minimal bleeding risk, but is often avoided if the patient is being considered for antithrombotic treatments such as tpa.
References
Sachs, C., Lehnhardt, M., Daigeler, A., Goertz, O. (2015). The Triaging and Treatment of Cold-Induced Injuries. Dtsch Arztebl Int., 112(44), 741-747. Doi:10.3238/arztebl.2015.0741
Petrone, P., Asensio, J., Marini, C. (2014). Management of accidental hypothermia and cold injury. Oct, 51(10):417-31. Doi: 10.1067/j.cpsurg.2014.07.004.
Imray, C., Grieve, A., Dhillon, S. (2009). Cold damage to the extremities: frostbite and non-freezing cold injuries. Postgrad Med J., Sep; 85(1007):481-8. Doi: 10.1136/pgmj.2008.068635
Pinaki Mukherji, MD
Assistant Professor
Department of Emergency Medicine
Department of Internal Medicine
Donald and Barbara Zucker School of Medicine at Hofstra/Northwell
References
Management of accidental hypothermia and cold injury. Petrone P, Asensio JA, Marini CP. Curr Probl Surg. 2014 Oct;51(10):417-31. doi: 10.1067/j.cpsurg.2014.07.004. Epub 2014 Jul 29.
Wilderness Medical Society practice guidelines for the out-of-hospital evaluation and treatment of accidental hypothermia: 2014 update.Zafren K1, Giesbrecht GG2, Danzl DF3, Brugger H4, Sagalyn EB5, Walpoth B6, Weiss EA7, Auerbach PS7, McIntosh SE8, Némethy M8,McDevitt M8, Dow J9, Schoene RB10, Rodway GW11, Hackett PH12, Bennett BL13, Grissom CK14.
Wilderness Environ Med. 2014 Dec;25(4 Suppl):S66-85. doi: 10.1016/j.wem.2014.10.010
Wilderness Medical Society Practice Guidelines for the acute treatment of Frostbite:2014 update. McIntosh SE1, Opacic M2, Freer L3, Grissom CK4, Auerbach PS5, Rodway GW6, Cochran A7, Giesbrecht GG8, McDevitt M9, Imray CH10, Johnson EL11, Dow J12, Hackett PH13. Wilderness Environ Med. 2014 Dec;25(4 Suppl):S43-54. doi: 10.1016/j.wem.2014.09.001.
Frostbite: a practical approach to hospital management. Handford C1, Buxton P2, Russell K3, Imray CE4, McIntosh SE5, Freer L6, Cochran A7, Imray CH8.Extrem Physiol Med. 2014 Apr 22;3:7. doi: 10.1186/2046-7648-3-7. eCollection 2014Gross, E., & Moore, J. (2012).
Using thrombolytics in frostbite injury. Journal of Emergencies, Trauma, and Shock, 5(3), 267. doi:10.4103/0974-2700.99709
Tintinalli, J. E., & Stapczynski, J. S. (2016). Tintinalli's emergency medicine: A comprehensive study guide. New York: McGraw-Hill.
Wrenn, K. (1991). Immersion foot. A problem of the homeless in the 1990s. Archives of Internal Medicine, 151(4), 785-788. doi:10.1001/archinte.151.4.785
Petrone, P., Kuncir, E., & Asensio, J. A. (2003). Surgical management and strategies in the treatment of hypothermia and cold injury. Emergency Medicine Clinics of North America, 21(4), 1165-1178. doi:10.1016/s0733-8627(03)00074-9
Gonzaga, T., Jenabzadeh, K., Anderson, C. P., Mohr, W. J., Endorf, F. W., & Ahrenholz, D. H. (2016). Use of Intra-arterial Thrombolytic Therapy for Acute Treatment of Frostbite in 62 Patients with Review of Thrombolytic Therapy in Frostbite. Journal of Burn Care & Research, 37(4). doi:10.1097/bcr.0000000000000245
How to Cite this Post
[Peer-Reviewed, Web Publication] Watts, S, Kaltman, D. (2020, Mar 16). Cold Management in the ED. [NUEM Blog. Expert Commentary by Mukherji, P]. Retrieved from https://www.nuemblog.com/blog/cold-injury
In-Flight Medical Emergencies
Written by: Andra Farcas, MD (PGY-3) Edited by: Phil Jackson, MD (PGY-4) Expert commentary by: William Brady, MD
In-Flight Medical Emergencies
You’re at 35,000 feet reclining in your tiny seat sipping your free ginger ale when suddenly: “Is there a medical professional on board?”
How often can you expect to encounter this?
The incidence of in-flight medical emergencies (IMEs) is difficult to accurately assess, as there is no mandated reporting and most studies gather data from ground medical support, which is not always contacted in flight. Studies estimate 1 in 604 flights will have an IME. Others report anywhere from 24-130 per 1 million passengers; since roughly 4 billion passengers fly annually, this means anywhere from 260-1420 emergencies daily worldwide. Thus, depending on how often you fly, there’s a good chance you’ll be called upon to volunteer.
What will you most commonly see?
The most common reason for an IME is syncope/pre-syncope, which make up about 33-37% of all IMEs. Other common complaints include GI symptoms (15%), respiratory symptoms (10-12%), cardiovascular complaints (7-8%), seizure or post-ictal state (6%), psychiatric (4%), allergic reaction (2%), suspected stroke (2%), diabetes complications (2%), obstetric emergencies (0.7%), and cardiac arrest (0.2-0.3%). About 4% of IMEs require a diversion of the flight.
How is the environment different?
Commercial air travel cabins are pressurized at 5000-8000ft elevation. At this pressure, gas expands by about 30%. Anatomical spaces containing gas, such as sinuses and middle ear, may be affected, as well as non-physiologic gas collections like a pneumothorax or post-operative air collections.
The partial pressure of oxygen is also low at this elevation so even a healthy individual will be mildly hypoxic to 90-93%.
The air is re-circulated, so passengers may be exposed to allergens even if they are many rows away. The re-circulated air is de-humidified so dehydration is also more likely.
Do you have to volunteer? Are you protected if you do?
Ethical obligation aside, do you have a legal obligation to volunteer? In the United States (as well as in Canada, England, and Singapore), physicians have no legal obligation to assist in an IME. In Australia, some European countries, and the Quebec province of Canada, however, they do.
If you do decide to volunteer, you are usually not required to have proof of your credentials in US flights but that may differ based on airline.
In the US, medical professionals who volunteer in IMEs are protected from liability by the Good Samaritan provision of the Aviation Medical Assistance Act except in cases of gross negligence or willful misconduct (such as intoxication, willfully harmful behavior, etc.). Note that if you seek compensation for helping, such as miles, seat upgrade, monetary, etc., you may jeopardize your standing under these immunity laws.
While airlines can get sued for conduct during IMEs, there has only been one reported case in the US of a medical professional being sued for volunteering to assist, and the case was dismissed without hearing.
What equipment and medications do you have?
In the US, all airlines must have the minimum required equipment, which includes basic supplies for assessment, airway/breathing, and intravenous access. Depending on the airline, you may find additional supplies as well.
Internationally, the International Civil Aviation Organization (ICAO), a United Nations agency, regulates international flight safety and recommends that “adequate” medical supplies be on board. While they have recommendations for what those supplies should be, these are non-mandatory and therefore the supplies will vary from country to country and airline to airline.
There are also minimum requirements for on board medications, and additional meds will vary based on airline and internationally.
What support do you have?
Flight attendants in the US are trained in CPR and operating the AED every 2 years. They are familiar with the supplies on board and can be a valuable resource.
If the IME is significant, you can also get assistance from medical ground support. These are medical professionals employed by companies who are contracted by airlines to help with IMEs. They are familiar with the equipment available on the flight, as well as medical resources at nearby airports. The flight crew will tell the pilot, who will contact the med support center and the airline operations center. The crew will then be your communication to ground med support. Since 45% of IMEs are responded to by flight crew alone (with the rest split between doctors and nurses/paramedics), the crew will likely consult ground med support before any interventions (meds, procedures) are taken.
What do you need to know about some specific scenarios?
Cardiac Arrest
Cardiac arrests are rarely the cause of IMEs (0.2-0.3%) but account for 86% of in-flight deaths. The crew should be trained in CPR and operating the AED so your time will be best spent on tasks like IV access and medication administration. Compression-only CPR is a reasonable option, although there are CPR masks on board if there are enough assistants. Terminating a resuscitation effort is tricky in an IME, but some authors suggest it is reasonable after 20-30 minutes of resuscitative effort without return of spontaneous circulation. Only a physician can pronounce death in flight.
Respiratory Compromise
Aircraft are not required to have oximeters. While there are oxygen tanks on US planes, they can only be set to low (2L/min) or high (4L/min) flow and the supply is limited.
COPD exacerbations are not uncommon and there should be bronchodilators on board.
It may be possible to request a descent to a lower altitude to improve oxygenation, although this is complicated given that lower altitude flight uses more fuel.
Pneumothoraces have been reported. If significant, there should be needles on board to perform needle thoracostomy. Descending to a lower altitude may also help in this instance with oxygenation and gas expansion.
Acute Coronary Syndrome
If ACS is suspected, there is aspirin 325mg on board which may be administered if no active bleeding or true allergy. While there is sublingual nitrogen on board, it should be used with caution, as there is no way to rule out an RV infarct.
Altered mental status
Glucometer availability may vary depending on the airline. A solution could be to ask other passengers, though this could compromise sterility and threaten patient privacy.
The seizure threshold may be lowered by in-flight hypoxemia and disturbance in circadian rhythms, and thus a high index of suspicion is advised. Suspected stroke is also one of the common reasons for diversion.
When is diversion reasonable?
The decision to divert a flight for a medical emergency is ultimately made by the captain of the aircraft with recommendations from ground med control and medical volunteers. There are many factors that go into consideration, including cost (anywhere from $20,000 to $725,000 has been reported), fuel amount (planes often take off with more fuel than it would be safe to land with), ability to land at the closest airport, and medical resources available at that airport. One study reported that even when there is a diversion for IME, only 1/3 of patients are actually taken to the hospital by EMS and only 1/3 of those transported are admitted. Most common causes for diversion include cardiac arrest, OB emergencies, cardiac symptoms, suspected stroke or other neuro symptoms, and respiratory symptoms.
Summary
IMEs can be daunting medical encounters to undertake given that you will be faced with limited diagnostic capabilities and a finite arsenal of medications and supplies to treat a wide variety of potentially life-threatening conditions. Regardless, we as emergency physicians are perhaps the most well-equipped medical professionals to handle this strenuous circumstance. If you provide reasonable care that meets your usual standards, then it is extraordinarily unlikely that you will ever be faced with liability. Always, be very cautious when accepting compensation as this may threaten your immunity. Remember that you are never alone and the decision to divert an aircraft will never rest on your shoulders without the support of the ground medical team.
Expert Commentary
Inflight medical emergencies (IME), an unanticipated medical event occurring on a commercial aircraft while in flight, represent a challenge to the volunteer healthcare provider.[1] The challenges are numerous, including the austere environment of the aircraft cabin, the almost complete absence of diagnostic studies, a very limited cache of medical supplies, and the distance from / time to definitive medical care. The aircraft cabin is cramped with little privacy and limited ability to place the patient in a comfortable, supine position; in addition, the ambient noise level is prohibitive to certain physical examination maneuvers, such as auscultation and blood pressure determination.
Diagnostic devices are largely non-existent; the most useful tool is your brain. Medications, basic supplies, and an automatic external defibrillator (AED) are present on all US commercial aircraft and many non-US airlines. While the AED is useful in the happily rare cardiac arrest, other supplies and medications are not particularly useful. In fact, the volunteer healthcare provider should not anticipate the presence of adequate supplies and medications…the aircraft is not an air ambulance or other medical facility. The Table lists required and additional medications and equipment. Please realize that the additional supplies are NOT present on US airlines for a range of reasons, most of which are appropriate and understandable. For example, if diazepam is included in the medical kit, significant risk for the airline is present regarding appropriate storage and use of this potent medication.
The volunteer healthcare provider, at times, may recommend an emergent landing. The volunteer healthcare provider should state his/her case with recommendations in basic, lay terminology. The decision to land the aircraft, however, is made by the captain of the vessel; should the captain disagree with the recommendation to urgently land the aircraft, do not argue. And also realize that during certain flights, there may not be an opportunity to divert and land…for instance, if the flight path is trans-Atlantic.
Lastly, as noted, volunteer healthcare provider on a US aircraft is protected from civil liability, assuming that he/she is not acting negligently in the approach to patient care.
Oddly, as I finished writing this commentary, on a flight to Munich, the announcement “Is there a doctor on board” interrupted my morning coffee. An adult female with a history of epilepsy was having a generalized tonic-clonic seizure. By the time that we (a paramedic and I) responded, she had stopped convulsing; she was post-ictal and gradually awakened over about 15 minutes. Our intervention was placing her in a safety position and maitianing an open airway during her initial posit-ictal period. By the time that we landed, she was alert without complaint. An ambulance met us at the gate; we provided a report on her condition and turned care over to the German medics. Afterwards, I documented the event in my own record and declined the airline’s offer of bonus frequent flyer miles.
References
Nable, J.V., Tupe, C.L., Gehle, B.D., Brady, W.J. In-Flight Medical Emergencies during Commercial Travel. NEJM. 2015;373(10):939-945
William J. Brady, MD
Professor of Emergency Medicine, Medicine, & Nursing
Department of Emergency Medicine
University of Virginia
How To Cite This Post
[Peer-Reviewed, Web Publication] Farcas, A. Jackson, P. (2020, Feb 17). In-Flight Medical Emergencies. [NUEM Blog. Expert Commentary by Brady, W]. Retrieved from http://www.nuemblog.com/blog/in-flight
Other Posts You Might Enjoy
References
Aerospace Medical Association Air Transport Medicine Committee. Medical Emergencies: Managing In-flight Medical Events (Guidance materials for health professionals). Aerospace Medical Association. 2016. http://www.asma.org/publications/medical-publications-for-airline-travel/managing-in-flight-medical-events.
Kodama, D., Yanagawa, B., Chung, J., Fryatt, K., Ackery, A.D. “Is there a doctor on board?”: Practical recommendations for managing in-flight medical emergencies. Canadian Medical Association Journal. 2018;26(190):E217-222
Martin-Gill, C., Doyle, T.J., Yealy, D.M. In-Flight Medical Emergencies: A Review. JAMA. 2018;320(24):2580-2590
Nable, J.V., Tupe, C.L., Gehle, B.D., Brady, W.J. In-Flight Medical Emergencies during Commercial Travel. NEJM. 2015;373(10):939-945
SCUBA Diving Injuries and Treatments
Written by: Aaron Wibberly, MD (NUEM PGY-2) Edited by: Sarah Dhake MD (NUEM Alum ‘19 ) Expert commentary by: Justin Hensley, MD
Expert Commentary
Thanks for writing about one of the topics that isn’t frequently covered in emergency medicine. SCUBA isn’t terribly dangerous, but early recognition of the problems encountered in these patients can save lives.
First, barotrauma exists because of Boyle’s Law (P1V1=P2V2). There is no way to go around this law of nature, only ways to prevent negative outcomes.
Barotrauma mainly affects the ears but can also affect the sinuses or mask.
Mask squeeze is failure to maintain pressure in facemask as pressure increases. Signs include subconjunctival hemorrhages, lid edema, skin ecchymosis, hyphema, and orbital hemorrhage (1)
Both tympanic membrane ruptures and inner ear problems (round and oval window) can be very disorienting underwater, placing the patient in danger of ascending too quickly or not being able to find their way out.
In an unresponsive diver, the arterial gas embolism is assumed until proven otherwise. Alveolar barotrauma is a life threat that needs emergent treatment.
Decompression sickness is known as “the bends” because of caisson workers. It mainly affected the hips and knees, causing them to maintain a bent over stance. For reasons nobody has yet identified, SCUBA divers are affected in the shoulders and elbows.
The “chokes” are bubbles in the pulmonary and cardiac vasculature, and can cause a “mill wheel” murmur (splash, splash, splash).
Skin bends, known as cutis marmorata, may be related to bubbles in the vasculature, but there is some evidence that it may be centrally mediated and symptomatic of more severe DCS. It frequently causes itching. (2,3)
Neurologic DCS most commonly affects the spinal cord and causes 50-60% of sport diver casualties.
Knowing where your closest hyperbaric chamber exists is of utmost importance. Diver’s Alert Network (DAN) does not publish a database but maintains a referral network that you can speak to after emergency evaluation by calling their 24-hour DAN Emergency Hotline at +1-919-684-9111.
References:
1. Barron, E. (2018). The “Squeeze,” an Interesting Case of Mask Barotrauma. Air Medical Journal, 37(1), 74-75. doi: 10.1016/j.amj.2017.10.003
2. Germonpre, P., Balestra, C., Obeid, G. and Caers, D. (2015). Cutis Marmorata skin decompression sickness is a manifestation of brainstem bubble embolization, not of local skin bubbles. Medical Hypotheses, 85(6), pp.863-869.
3. Kemper TC, Rienks R, van Ooij PJ, and van Hulst RA. (2015). Cutis marmorata in decompression illness may be cerebrally mediated: a novel hypothesis on the aetiology of cutis marmorata. Diving Hyperb Med., 45(2), pp. 84-8.
Justin Hensley, MD
Founder and Editor of EBM Gone Wild
Quality Improvement/Assurance Director,
CHRISTUS Health-Texas A&M-Spohn Emergency Medicine Residency
How to Cite this Post
[Peer-Reviewed, Web Publication] Wibberly A, Dhake S. (2019, Aug 27). SCUBA Diving Injuries and Treatments. [NUEM Blog. Expert Commentary by Hensley J]. Retrieved from http://www.nuemblog.com/blog/scuba.
Other Posts You Might Enjoy
Sink or Swim: The management of submersion injuries
Written by: Michael Conrardy , MD (NUEM PGY-2) Edited by: Gabby Ahlzadeh, MD (NUEM ‘19) Expert commentary by: Kristina McAteer, MD
Introduction
If you are an emergency physician, you likely will see submersion injuries often throughout your career. In the United States, from 2005-2014, there are approximately ten deaths per day from unintentional drowning, and about 3,536 annually. This number increases to eleven per day if you include boating-related deaths. One in five deaths involve children under the age of fourteen, and drowning is the second leading cause of death for children ages 1-14 [1,2]. Approximately 80% of all victims are male [2], and when considering adolescents and adults, alcohol is involved in up to 70% of cases [3]. Statistics for nonfatal drowning events are difficult to obtain, but studies estimate that they may occur several hundred times more frequently than reported deaths [4,5].
Example Case
While working in the emergency department on Friday night, three college students are brought in by EMS after “drinking alcohol and jumping in Lake Michigan.” Patient #1 received CPR for ten minutes en route, has a temperature of 32 degrees Celsius, signs of head trauma, and a cervical collar in place. Patient #2 experienced a short period of submersion and is in mild respiratory distress. Patient #3 was submerged for only a brief period of time and is completely asymptomatic.
Emergency Department Management of Submersion Injuries
Patient #1 - Severely Symptomatic Patients
Initial Management:
In serious cases of drowning, top concerns are hypoxemia, hypothermia, subsequent cardiac arrest, and other signs of trauma.
If in cardiac arrest, start ACLS and consider underlying causes such as hypoxemia or hypothermia
Remove wet clothing and use rewarming techniques
Chest x-ray, blood gas, labs, end tidal CO2. Initial CXR is often normal but can be helpful in tracking the patient’s condition.
Assess for signs of trauma and obtain appropriate imaging. It is important to note that cervical spine injuries are uncommon (<5%) in cases of drowning, and cervical collars should not distract from airway management [6].
Management of airway, ventilation, oxygenation:
Fluid aspiration can lead to loss of surfactant, pulmonary edema, and hypoxemia from V/Q mismatch. Hypoxemia is the primary cause of end-organ damage in submersion injuries (typically cardiac and neurologic) and treating it should be your top priority. Here are some general guidelines:
Airway: Intubate patients if unable to protect airway, O2Sat < 90% despite supplemental O2, or PaCO2 >50
Ventilation: Positive pressure opens alveoli and improves ventilation in drowning.
Unable to protect airway: Start with bag-valve-mask and then intubate. Use ARDS settings of Vt 6-8 mL/kg, Pplateau < 30mmHg, and increased PEEP.
Able to protect airway: Use non-invasive positive pressure ventilation in alert, symptomatic patients for goal O2Sat > 94% [6].
Oxygenation: Use maximum available supplemental O2 initially and wean as tolerated.
Medications:
No medications need to be routinely used for submersion injuries. Bronchospasm can occur, and if suspected should be treated with inhaled beta-adrenergic agonists. Glucocorticoids have not been shown to be helpful in preventing subsequent lung injury and may interfere with healing, and not enough data exists to support routine administration of exogenous surfactant. Antibiotics should only be used in cases where signs of infection develop or if the patient was submerged in grossly contaminated water [7].
ECMO:
Data on use of ECMO to treat patients with severe pulmonary edema or signs of ARDS are extremely limited, although initial results are encouraging [9]. This is an area of ongoing research, and perhaps we may eventually see early initiation of ECMO as standard practice for severely symptomatic patients.
Patient #2 - Mildly Symptomatic Patients
The main takeaway from patient #2 is that all patients who are symptomatic on arrival to the ED (requiring ventilatory assistance or supplemental O2) should be admitted to the hospital. Airway, ventilation, and oxygenation should be managed as above, and serial chest x-rays can help monitor for development of pulmonary edema in symptomatic patients [6].
Patient #3 - Asymptomatic Patients
Although most research on safe discharge has been in children, it is generally accepted that asymptomatic patients can be safely discharged after 4-6 hours of monitoring. Patients should have normal pulmonary exam, SaO2>95% on room air, and GCS>13 [6,10]. Some clinicians advocate for observing up to eight hours based on a study that observed one pediatric patient who did not develop symptoms until seven hours post-submersion [11], although six hours in the ED with return precautions is probably adequate in most cases. Many clinicians obtain basic labs on all patients and a chest x-ray prior to discharge to confirm that there are no signs of pulmonary edema, although no strong evidence supports these practices.
Key Takeaways:
Hypoxemia is the main cause of harm, prioritize the airway and use positive pressure
Initial chest radiographs are often normal, but can be used to track a patient’s clinical status
Blood gas is the most useful laboratory test in symptomatic patients, additional testing (e.g. ECG, CBC, chem, troponin) is particularly useful in patients with altered mental status
Asymptomatic patients may be safely discharged after six hours of monitoring
Expert Commentary
The terminology can be confusing with such terms as drowning, nonfatal drowning, submersion injury, dry drowning, wet nonfatal drowning, and so on. Most recently (2010) the AHA recommended a more straightforward approach and supported the Utstein definition which hopefully offers some clarification, relieves some of the angst when approaching this population, and standardizes research and reporting. The Utstein defines a drowning as “a process resulting in a primary impairment from submersion or immersion in a liquid medium.” [1]
The key is to keep it simple. Is your patient symptomatic from the exposure? If so, focus on the ABC’s and get to work!
You will often see pediatric patients as drowning victims. Drowning is a leading cause of accidental death in children under 5 years of age, particularly in states where swimming pools or beaches are more accessible. The states of California, Arizona and Florida lead the way [2]. The second peak is in older ages (15-25 yo), primarily males, and involves more rivers, lakes and beaches. [3]. The distinction between fresh and salt water drownings is no longer considered important as the volume of aspirated water is so small that the theorized electrolyte shifts do not have any real physiological impact [4]. Both wash out pulmonary surfactant.
As appropriately pointed out it is often is not the drowning that causes mortality. It is therefore necessary to consider concomitant trauma, toxic ingestions (at the very least alcohol leading to impaired judgment!), and hypothermia. Hypoxemia and the resultant hypoxia is ultimately the cause for death, often impacting multiple organ systems, and thus it is your job to reverse the hypoxemia as quickly as possible! Supplemental oxygen, BIPAP (Bilevel positive airway pressure), endotracheal intubation and if necessary, ECMO (extracorporeal membrane oxygenation) are management options. There is a neuroprotective effect of hypothermia and in some cases patients make a complete recovery despite a prolonged resuscitation. Age > 14, duration of submersion > 5 minutes, time to basic life support > 10 minutes, resuscitation duration > 25 minutes, GCS (Glascow coma scale) <5, persistent apnea and requirement of CPR in the emergency department and arterial blood pH <7.1 have all been associated with a poor prognosis. [5]
Vanden Hoek TJ, Morrison LJ, Shuster M, et al. Part 12: Cardiac Arrest in special situations 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergent Cardiovascular Care. Circulation 2010; 122: S829.
Brenner Ra. Prevention of drowning in infants, children, and adolescents. Pediatrics 2003; 112:440.
DeNivola, LK, Falk, JL, Swanson ME, et al. Submersion injuries in children and adults. Crit Care Clin 1997;12:477.
Orlowski JP, Szpilman D. Drowning, Resucue, resuscitation and reanimation. Pediatr Clin. North Am 2001. 48:627.
Quan L, Mack CD, Schiff MA. Association of water temperature and submersion duration and drowning outcome. Resuscitation 2014.85:790.
Kristina McAteer, MD
Assistant Professor of Emergency Medicine
Brown University
How To Cite This Post
[Peer-Reviewed, Web Publication] Conrardy M, Ahlzadeh G. (2019, July 1). Sink or Swim: The management of submersion injuries. [NUEM Blog. Expert Commentary by McAteer Kristina]. Retrieved from http://www.nuemblog.com/blog/drowning.
Other Posts You May Enjoy
References
Centers for Disease Control and Prevention, National Center for Injury Prevention and Control. Web-based Injury Statistics Query and Reporting System (WISQARS) [online]. Available from: URL: http://www.cdc.gov/injury/wisqars.
CDC. Wide-ranging online data for epidemiologic research (WONDER). Atlanta, GA: CDC, National Center for Health Statistics; 2016 [online]. Available at http://wonder.cdc.gov.
Driscoll TR, Harrison JA and Steenkamp M. Review of the role of alcohol in drowning associated with recreational aquatic activity. Inj Prev. 2004; 10: 107-13.
Bierens JJ, Knape JT and Gelissen HP. Drowning. Curr Opin Crit Care. 2002; 8: 578-86.
Orlowski JP. Drowning, near-drowning, and ice-water drowning. JAMA : the journal of the American Medical Association. 1988; 260: 390-1.
Schmidt AC, Sempsrott JR, Hawkins SC, Arastu AS, Cushing TA and Auerbach PS. Wilderness Medical Society Practice Guidelines for the Prevention and Treatment of Drowning. Wilderness Environ Med. 2016; 27: 236-51.
Layon AJ and Modell JH. Drowning: Update 2009. Anesthesiology. 2009; 110: 1390-401.
Chandy D, Weinhouse GL. Drowning (submersion injuries). UpToDate. Danzl DF, Grayzel, J (Ed). 2017.
Burke CR, Chan T, Brogan TV, et al. Extracorporeal life support for victims of drowning. Resuscitation. 2016; 104: 19-23.
Causey AL, Tilelli JA and Swanson ME. Predicting discharge in uncomplicated near-drowning. The American journal of emergency medicine. 2000; 18: 9-11.
Noonan L, Howrey R and Ginsburg CM. Freshwater submersion injuries in children: a retrospective review of seventy-five hospitalized patients. Pediatrics. 1996; 98: 368-71.
Nibbles and Bits: Management of Dog Bite Wounds
The classic teaching is that bite wounds should not be closed due to concern for infection; however, several studies have challenged this dogma, particularly in low-risk wounds and patients. We discuss the evidence this week on the blog.



























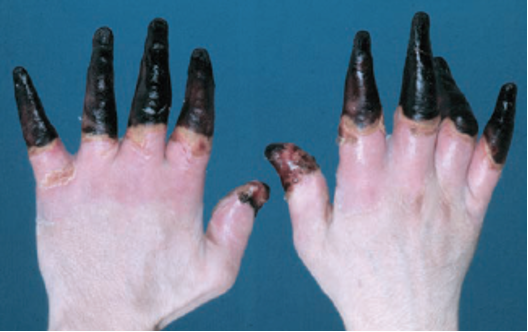

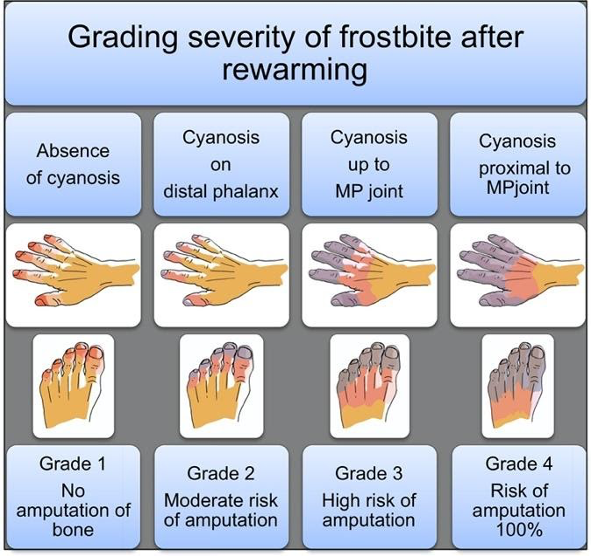
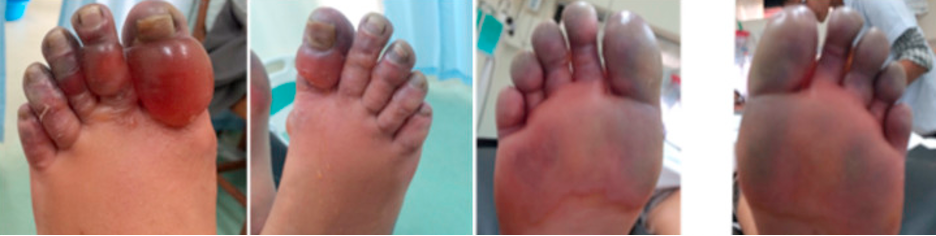
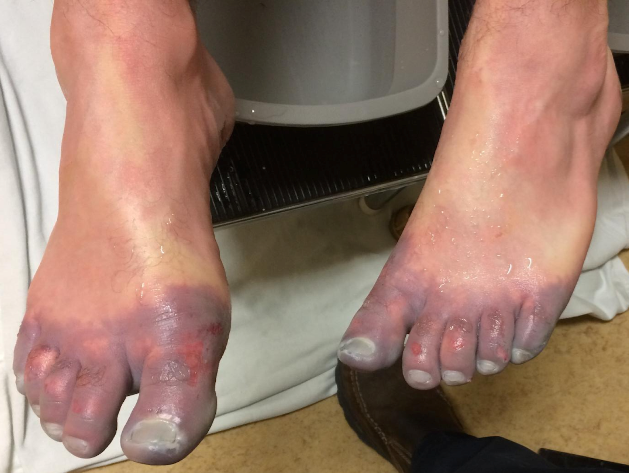
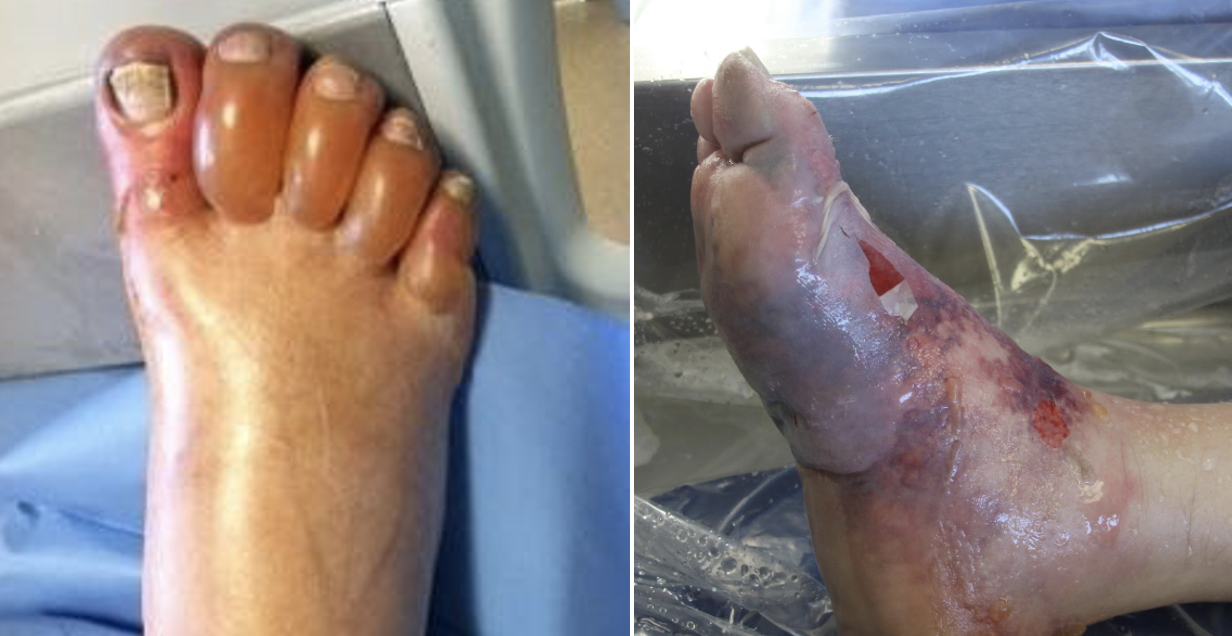


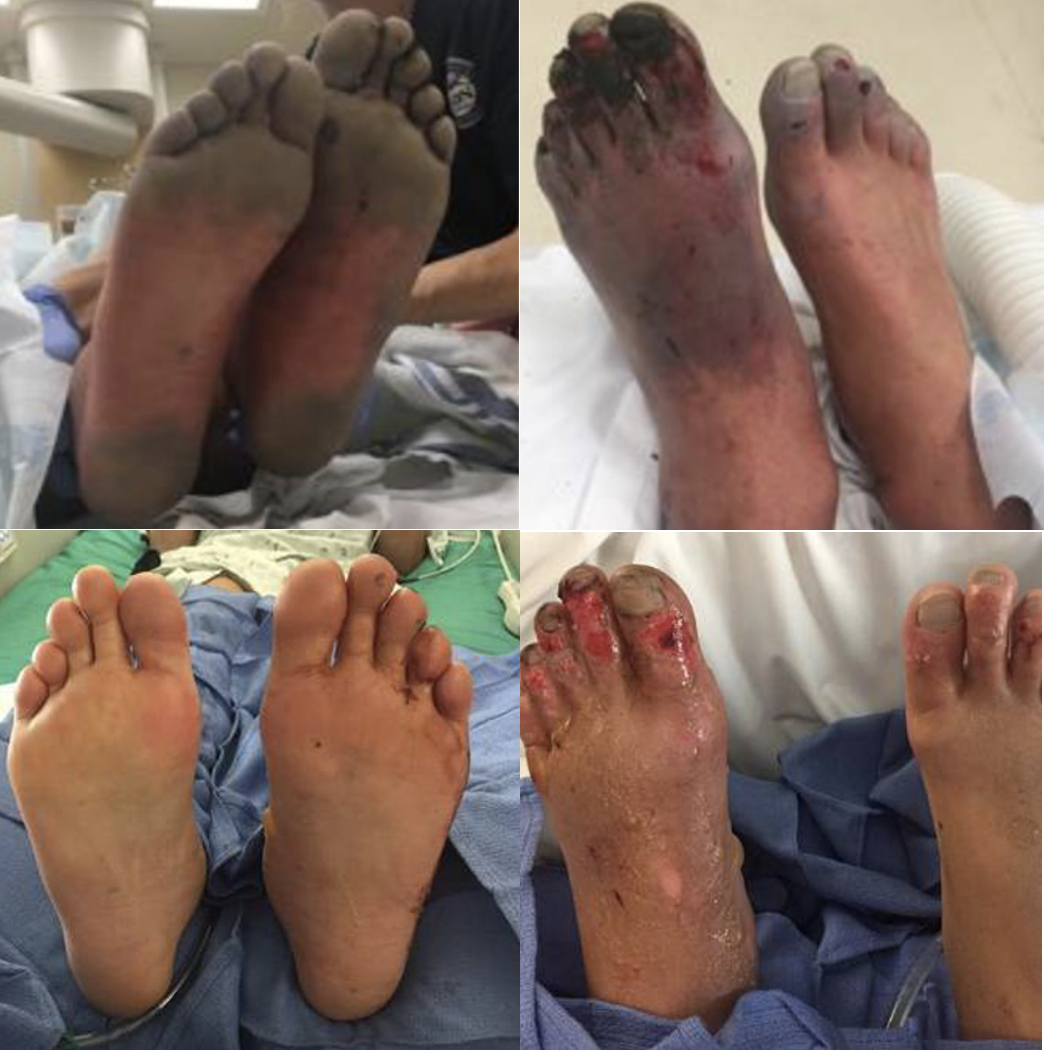
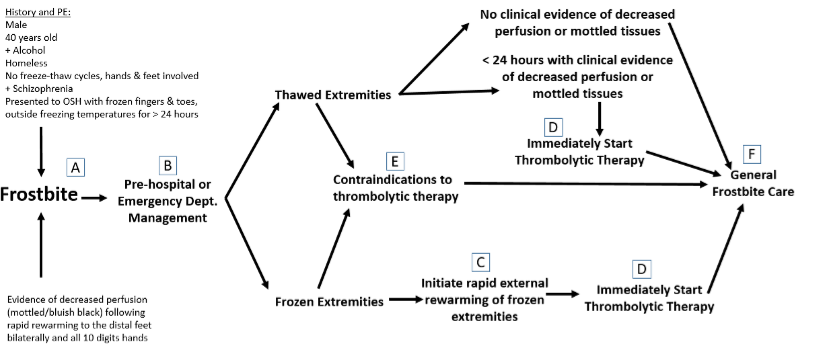




![Summary of the Pathophysiology of Heat Stroke [1]](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1593785361684-SVDHAE847O881FLLW493/Picture1.png)
![Subject in a cold water-immersion bath after heat- stroke [4]](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1593785609566-OWN8IJYW5Y4QGFO2YPWI/Picture2.png)
![Evaporative and conductive cooling methods--note the placement of ice packs in axilla, groin as well as the cooling fan overhead [4]](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1593785678671-8PWD8653TJQ61EQ1F6VS/Picture3.png)
![table of cooling methods [6]](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1593785810930-ZUZ2S1CGXXO1ME2854ZJ/Picture4.png)
![Collapse_Algorithm[1] (1) (1).png](https://images.squarespace-cdn.com/content/v1/549b0d5fe4b031a76584e558/1593786307567-LB1EFX5KMHS371YBFKMZ/Collapse_Algorithm%5B1%5D+%281%29+%281%29.png)